Reviews and Information for Control Solutions Inc.
Search term may appear only in full report available to members. Join now for full access.
Product Review
Low-Dose Lithium Supplements Review
Choose the Best Low-Dose Lithium Supplement. CL Tests Reveal Which Low-Dose Lithium Supplements Offer the Best Quality and Value.

Product Review
Toprol XL vs. Generic Metoprolol Succinate Extended-release (ER) Tablets Review Article
Choose the Best Blood Pressure Medication. Find Out Why Some Generic Blood Pressure Medications Are Not the Same as the Original & May Increase Blood Pressure or Have Other Disturbing Side Effects.
Product Review
Ginger Supplements, Chews & Spices Review
Tests Reveal Best and Worst Ginger Supplements & Spices. Poor Quality and Lead Contamination Discovered in Some Products.

Product Review
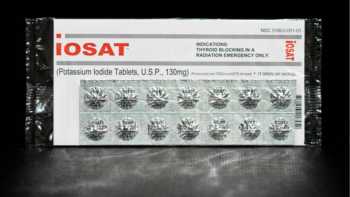
Potassium Iodide (KI) and Iodate (KIO3) Radioprotective Pills Review
Learn How to Choose the Best Radioprotective Iodine Pill and Use It Properly
Product Review
Garlic Supplements Review
Find the Best Garlic Supplements. CL Tests Reveal Big Differences in Garlic Strength -- Some Have Little to No Garlic!.

Product Review
L-Lysine Supplements Review
Read L-Lysine Labels Carefully. Some Can Fool You.
Product Review
Green Coffee Bean Extract Supplements Review (for Weight Loss)
Choose the Best Green Coffee Bean Extract. 50% of Green Coffee Bean Extract Supplements Don't Deliver Expected Ingredients.

Product Review
Weight Loss Supplements Review (7-Keto DHEA, Forskolin and Stimulant Blend Supplements)
Choose the Best Weight Loss Supplement. Be Careful With Weight Loss Supplements — Few Pass Quality Testing and Safety Review.

CL Answer
What is hydrogen water? Is it beneficial for alertness, athletic performance, arthritis or other conditions, and is it safe?
Find out if hydrogen water has benefits with regard to alertness, side effects of radiotherapy in cancer patients, metabolic syndrome, athletic performance, rheumatoid arthritis, liver disease, and other conditions, and learn if it is safe to use.

CL Answer
Dimethyl sulfoxide (DMSO): Health Effects & Safety Concerns
Dimethyl sulfoxide (DMSO) is a colorless liquid promoted for osteoarthritis, interstitial cystitis, tennis elbow, rotator cuff tendonitis, wound healing, and many other conditions. Find out if it works and if it's safe.

CL Answer
Does AZO Bladder Control really work for overactive bladder?
AZO Bladder Control information, including results from clinical studies on bladder control, dosage, and safety.

CL Answer
What are the best bug sprays to prevent tick and mosquito bites? Is the insecticide permethrin safe to use?
Find out how permethrin sprays differ and which can be used on sheets and sleeping bags versus clothing

CL Answer
Does AHCC or active hexose correlated compound have anticancer or antiviral benefits, and is it safe?
Learn about AHCC and active hexose correlated compound, including whether these compounds are beneficial for liver, pancreatic, or prostate cancer, chemotherapy-related side effects, or the flu. Also, find out if AHCC or active hexose correlated compound are safe.

CL Answer
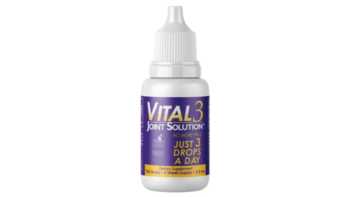
Do Vital 3 collagen drops reduce joint pain?
Learn more about Vital 3 collagen drops withundenatured type II collagen, and if they can help joints move better and feel better.
CL Answer
LifeWave Patches: Do They Work?
LifeWave patches are promoted for numerous health benefits, but does research support these claims? Find out, and learn about possible safety concerns, cost, and controversies related to LifeWave patches.
CL Answer
What are phytoceramides? Do phytoceramide supplements really work to improve aging skin?
Phytoceramide supplement information, including what they are, what they claim to do, and if they have anti-aging effects.

CL Answer
Can I trust lab reports and Certificates of Analysis provided by supplement manufacturers?
Lab reports and Certificates of Analysis are provided by supplement manufacturers, but sometimes include incorrect testing and "dry labbing". ConsumerLab.com's answer explains.

CL Answer
Akkermansia muciniphila: Health Benefits and Safety
Akkermansia muciniphila has gained attention for its potential benefits in managing insulin resistance, diabetes, weight loss, cancer, and other health conditions, but does it really work, and is it safe? Find out.

CL Answer
Is ingesting colloidal silver helpful for any condition and is it safe to use?
Find out if there is evidence showing colloidal silver can help to treat infections, boost the immune system, help with arthritis and more, plus find out if colloidal silver can cause blue skin or other side effects.

CL Answer
Do hair loss supplements, such as Viviscal, Hair La Vie, and Nutrafol, or topical essential oils work?
Vitamins and supplements that may help with hair loss and thinning, including saw palmetto, beta-sitosterol, protein, iron, and vitamin D.

CL Answer
Do any supplements help with COVID-19? Do supplements like vitamin D, zinc, vitamin C, or herbals work?
Find out if natural remedies & supplements for coronavirus such as zinc, vitamin C, garlic, or elderberry help to prevent or treat COVID-19.
CL Answer
What is the ingredient zeolite I see in Natural Cellular Defense and other detox supplements? Does it help in any way?
Learn what zeolite is and why it is found in many detox supplements like Waiora's Natural Cellular Defense, Get Healthy Again Zeolite, and Ultra Liquid Zeolite.

Clinical Update
6/30/2023
Vital 3 for Joint Pain?
Does Vital 3 Joint Solution reduce joint pain? Find out in our updated article about Vital 3 Joint Solution.
CL Answer
What are the health benefits of grapefruit seed extract (GSE)? Is it safe?
Learn more about grapefruit seed extract health benefits, including its possible antifungal and antibacterial effects and its safety.
CL Answer
Are fruit and vegetable “washes” or devices more effective than water for cleaning produce and removing pesticides? Are they safe?
Fruit and veggie washes, vinegar, and baking soda, as well as devices that use electrolysis, ozone water, or ultrasonic cleaning, are often promoted for removing dirt, bacteria and pesticides from produce. Find out if any work better than rinsing with water and if they are safe.

CL Answer
Which supplements and foods can help lower or control blood sugar?
Find out which supplements can help lower or control blood sugar. Supplements including turmeric/curcumin, fiber, cinnamon, and ginseng are believed to help lower blood sugar.
CL Answer
Do any supplements help with gum disease or periodontitis?
Supplements for healthy gums may help with gum disease or periodontitis. Learn more about omega fatty acids, lycopene, and probiotics.

CL Answer
Can I use a home test for mercury to check for mercury in fish, like canned tuna, fresh fish, and sushi?
Find out if home mercury test kits can be used to test fish, such as canned tuna, fresh fish or sushi for mercury contamination. Plus, find out how much mercury ConsumerLab found in popular canned tuna and canned salmon. ConsumerLab.com's answer explains.

Clinical Update
10/16/2025
Constipation Solutions: What Works. What Doesn’t
Experts have weighed in on the best remedies for chronic constipation. Find out which foods and supplements made the cut:
Recalls & Warnings
July 23, 2025
FDA Warns Seller of Allegro Eye Drops, Tea Tree Oil Eyelid Wipes, and More
On July 9, 2025, the FDA issued a Warning Letter to Scope Health Inc.
Recalls & Warnings
May 08, 2023
SD Biosensor Pilot At-Home COVID-19 Tests Recalled Due to Bacterial Contamination
On May 4, 2023, the FDA warned consumers and health care providers not to use certain lots of Roche Diagnostics’ SD Biosensor, Inc. Pilot COVID-19 At-Home Tests due to bacterial contamination in the test kit’s liquid solution.
Recalls & Warnings
June 10, 2024
StellaLife Homeopathic Oral Rinse & Spray Recalled Due to Yeast, Mold, and Bacteria
On June 5, 2024, HomeoCare Laboratories, Inc.
Recalls & Warnings
December 19, 2024
Jarritos Coconut Water Recalled
On November 4, 2024, Tipp Distributors, Inc. issued a recall all lot codes of Jarritos Coconut Water (17.5 fl oz cans) because the airtight seal on the lid of the cans may be compromised.
Recalls & Warnings
May 04, 2023
FDA Warns MedoLife for Promoting Homeopathic Products to Treat COVID-19, Cancer
On April 19, 2023, the FDA issued a warning letter to Medolife Rx D/B/A MedoLife Corp., AELIA Inc. and Queanta Inc.
Recalls & Warnings
January 23, 2025
Glutathione Distributor Warned by FDA
On November 13, 2024, the FDA issued a Warning Letter to Western Innovations, Inc.
Recalls & Warnings
May 08, 2024
Razer, Inc. to Pay Over $1.1 Million Settlement for Zephyr Face Mask ”N-95” Claims
On April 29, 2024, the FTC announced it will be returning over $1.1 million to consumers who purchased Zephyr face masks after the company promoted its Zephyr face masks as N95-grade despite never submitting for testing to the FDA for approval of such claim.
Recalls & Warnings
January 13, 2026
Sea Moss Gel Superfood Recalled Due to Possible Health Risk
On January 9, 2026, Diva Fam Inc.
News Release
December 06, 2016
Which Brands of Alpha Lipoic Acid are Best? -- ConsumerLab.com Tests Lipoic Acid Supplements for Quality, Identifying Top Picks
White Plains, New York, December 6, 2016 — Alpha-lipoic acid supplements may improve insulin sensitivity and blood sugar control in people with type 2 diabetes, reduce symptoms of diabetic peripheral neuropathy, and enhance weight loss when dieting.
Recalls & Warnings
March 30, 2023
Seller of Tollovid Supplements Warned for COVID-19 Claims
On November 7, 2022, the FDA issued a warning letter to Todos Medical Ltd, aka Todos Medical USA Inc, following a review of the company’s websites, social media, and Amazon storefronts, which found statements about the company’s Tollovid 3CL Protease Inhibitor Delayed Release ...
Recalls & Warnings
September 14, 2023
FDA Warns CVS, Walgreens, Similasan & Others for Eye Drop Violations
On September 11, 2023, the FDA issued Warning Letters to the following eight sellers of homeopathic and other types of eye drops regarding the products noted in italics due to a variety of violations of FDA regulations, most notably that they were marked with claims suggesting that they could cure, ...
Recalls & Warnings
October 06, 2020
FDA Warns Seller of Red Yeast Rice, Vitamin D, Blood Pressure Supplements, and More
On August 28, 2020, the FDA issued a warning letter to Dr. Sam Robbins, Inc.
Recalls & Warnings
September 21, 2023
Moor Herbs, Inc Warned for Manufacturing Violations, Drug Claims
On August 1, 2023, the FDA issued a Warning Letter to Moor Herbs, Inc.
Recalls & Warnings
April 24, 2024
NOW Solutions XyliWhite Coconut Oil Toothpaste Gel Recalled
On March 6, 2024, NOW Foods issued a recall of over 27,000 units of NOW Solutions XyliWhite Coconut Oil Toothpaste Gel because it has an off taste and flavor.
Recalls & Warnings
August 28, 2023
Dr. Berne’s MSM and Castor Oil Eye Drops Recalled Due to Bacterial and Fungal Contamination
On August 26, 2023, Dr. Berne’s Whole Health Products issued a recall of all lots of its MSM DROPS 5%, 15% Solution, Dr. Berne’s Organic Castor Oil Eye Drops and Dr. Berne’s MSM MIST 15% Solution due to the risk of bacterial and fungal contamination.
Recalls & Warnings
July 17, 2023
Seller of Ashwagandha, Lion’s Mane & Other Supplements Warned for Manufacturing Violations
On March 17, 2023, the FDA issued a warning letter to Brand Packaging Group, Inc. after FDA inspection of the company’s facility found products to be adulterated because they were prepared, packed, or held under conditions that do not meet Current Good Manufacturing Practices (CGMP).
Recalls & Warnings
June 16, 2020
FDA Warns Four Companies for Unsafe "Homeopathic" Injectables
On June 16, 2020, the FDA issued warning letters to four manufacturers of unapproved injectable drugs labeled as homeopathic.
Recalls & Warnings
December 01, 2022
FTC Takes Action Against Company Promoting “COVID Resist” Supplement to Treat COVID-19
On November 22, 2022, the FTC filed a complaint in a U.S. district court against California-based company Precision Patient Outcomes, Inc. for promoting its COVID Resist and VIRUS Resist supplements to prevent and treat COVID-19.
Recalls & Warnings
May 09, 2022
Natural Organics Keto Capsules Recalled Due to Gluten
On March 6, 2022, Natural Organics, Inc. issued a recall of four lots of NaturePlus Keto Living Sugar Control Capsules that were found to contain gluten.
Recalls & Warnings
March 11, 2024
Healthex Warned for Manufacturing Violations
On June 15, 2023, the FDA issued a Warning Letter to Healthex Distributors, Inc.
Recalls & Warnings
October 11, 2023
Family Sentenced to Over 12 Years in Prison for Selling Dangerous “Bleach” Miracle Mineral Solution as “COVID Cure”
Four Florida men who distributed the toxic bleach solution Miracle Mineral Solution (MMS) as a cure for COVID-19 and other serious medical conditions have been sentenced to 5 to 12 years in prison for conspiring to defraud the United States.
Recalls & Warnings
January 04, 2024
Toxic Herb Found in More Tejocote Root Supplements
On January 3, 2024, the FDA warned consumers not to purchase or use certain tejocote root supplements after FDA laboratory analysis confirmed the products contain yellow oleander (Thevetia peruviana), a toxic herb.
Recalls & Warnings
February 05, 2025
Muscle Tech Alpha Test Recalled
On December 18, 2024, Iovate Health Sciences USA Inc. recalled 10 lots (163,248 units) of Muscle Tech Alpha Test capsules because they contain cathine, a controlled substance that can cause serious adverse effects.
News Release
October 02, 2008
ConsumerLab.com finds melatonin supplements range in strength but ingredient quality is high
WHITE PLAINS, NEW YORK — OCTOBER 2, 2008 — ConsumerLab.com announced test results today from its new Product Review of Melatonin Supplements. Melatonin supplements may help treat certain sleep disorders and disturbances, including jet lag.
News Release
March 13, 2006
ConsumerLab.com reports on supplements for bone health containing calcium and vitamin D— Results posted for 32 supplements for adults and children; One found contaminated with lead
WESTCHESTER COUNTY, NEW YORK — MARCH 13, 2006 — ConsumerLab.com announced test results today from its new Product Review of Supplements for Bone Health covering 32 adult and children's products containing calcium and vitamin D. Sales of calcium supplements in the U.S.
News Release
July 10, 2002
Pharmavite dietary supplements receive ConsumerLab.com approval — Vitamin E, SAM-e, St. John's Wort, Ginkgo and others merit approved quality products ranking
WHITE PLAINS, NY — July 10, 2002 — ConsumerLab.
Recalls & Warnings
September 10, 2021
FDA Warns Ten Sellers of "Diabetes" Supplements
On September 7, 2021, the FDA issued warning letters to 10 supplement companies that made drug claims by promoting products to treat diabetes and/or lower blood sugar. Five of the products were sold on Amazon as well as on company websites. The products were promoted with statements such as
Recalls & Warnings
October 19, 2023
Jay Robb Protein Powder Recalled
On October 13, 2023, Select Custom Solutions recalled one lot of Jay Robb Vanilla Flavored Egg White Protein 24 oz because the product may contain hard, plastic, foreign material which could pose a choking hazard. No adverse events have been reported to date.
Recalls & Warnings
January 04, 2024
Lone Star Botanicals Warned by FDA for Manufacturing Violations
On November 26, 2023, the FDA issued a Warning Letter to Lone Star Botanicals, Inc.
Recalls & Warnings
September 01, 2022
Elite One Source Nutritional Services Warned for Manufacturing, Labeling Violations
On August 5, 2022, the FDA issued a warning letter to dietary supplement manufacturer Nutritional Laboratories International, Inc. (DBA Elite One Source Nutritional Services, Inc.
Recalls & Warnings
December 29, 2022
EarthLab, Inc. Warned for Promoting Curcumin, Elderberry to Treat Pain & Flu
On November 10, 2022, the FDA issued a warning letter to EarthLab, Inc., dba Wise Woman Herbals following inspection of the company’s website which found statements about the company’s products to be drug claims because they suggest the products can prevent or treat disease.
Recalls & Warnings
August 16, 2023
FDA Warns Sun Ten Laboratories for Manufacturing Violations, Drug Claims
On April 7, 2023, the FDA issued a warning letter to STPCA Inc. dba Sun Ten Laboratories because products were found to be adulterated because they were prepared, packed, or held under conditions that do not meet Current Good Manufacturing Practices (CGMP).
Recalls & Warnings
March 13, 2020
FDA Finds Problems at 52% of Supplement Manufacturing Sites in U.S. and 42% Abroad
ConsumerLab.com has obtained results of the FDA's inspections in Fiscal Year 2019 (October 1, 2018 - September 30, 2019) of 598 dietary supplement manufacturing facilities in the U.S.
Recalls & Warnings
March 09, 2022
FDA Warns Seller of Magnesium, CBD, Herbal Extracts & More
On February 9, 2022, the FDA issued a warning letter to Bea Lydecker’s Naturals, Inc.
Recalls & Warnings
August 17, 2022
Seller of Moringa Tea and Other Herbal Products Warned for Drug Claims, Manufacturing Violations
On July 29, 2022, the FDA issued a warning letter to Deggeh Foods, Inc.
Recalls & Warnings
March 09, 2022
Court Bars Salud Natural From Selling Aloe, Joint Supplements & More
On March 8, 2022, a federal court ordered Salud Natural Entrepreneur, Inc. to stop distributing nutritional supplements that violate the Federal Food, Drug and Cosmetic Act (FDCA).
Recalls & Warnings
July 18, 2022
CVS, Target, Walgreens, and Other Oral Magnesium Citrate Laxatives Recalled Due to Bacterial Contamination
On July 14, 2022, Magnesium Citrate Laxative Oral Solution Lemon Flavor products manufactured by Vi-Jon, LLC and sold under various store brand names were recalled after testing identified the presence of the bacteria Gluconacetobacter liquefaciens.
Recalls & Warnings
March 16, 2023
Omega-3 Supplements for Dogs and Cats Recalled Due to Potential for Vitamin A Toxicity
On March 9, 2023, Stratford Care USA issued a recall of 62 brands of omega-3 supplements for dogs and cats due to potentially elevated levels of vitamin A.
Recalls & Warnings
May 08, 2024
APG SEVEN Warned for Promoting Products for Cancer, Depression, Arthritis, & More
On April 18, 2024, the FDA issued a Warning Letter to APG SEVEN, INC following a review of the company’s product labels, website, and social media, which found statements about the company’s BioMoringa, BioDiabetin, Maca Plus, BioProstate, Immune Support, HongoTrap, Honbacterol, ...
Recalls & Warnings
April 27, 2023
Seller of Magtein, Veggie Caps, & More Warned for Drug Claims, Manufacturing Violations
On March 8, 2023, the FDA issued a warning letter to Spartan Enterprises Inc., dba Watershed Wellness Center, after a review of the company’s website found statements about its Dr. Bob’s Naturals Spirulina, Dr. Bob’s Naturals Magtein, Dr.
Recalls & Warnings
April 03, 2024
FDA Warns Ambaya Gold for Promoting Products for Depression, Cancer, & Arthritis
On December 5, 2023, the FDA issued a Warning Letter to Ambaya Gold Health Products, LLC following review of the company’s website and social media, which found statements about the company’s Brain Balance, Immune System Boost, Dentist In A Bottle, Essensiac, Fulvic Green, Silver ...
Recalls & Warnings
October 16, 2025
More Ground Cinnamon Found to Contain Lead
On October 10, 2025, the FDA warned consumers not to consume three brands of ground cinnamon, which were found to contain elevated levels of lead. This brings the total to 16 brands that the agency has included in its public health alert about lead in cinnamon.
Recalls & Warnings
June 20, 2025
Yerba Prima Great Plains Bentonite + Herbal Detox Capsules Recalled
On June 10th, 2025, Yerba Prima Inc recalled 4,985 bottles of Great Plains Bentonite & Herbal Detox Capsules due to potential contamination with Pseudomonas aeruginosa.
Recalls & Warnings
June 09, 2025
Nordic Naturals Zero Sugar Multivitamin Gummies for Kids Recalled
On May 2, 2025, Nordic Naturals, Inc. recalled 1,164 bottles of Nordic Naturals Zero Sugar Kids Multivitamin Supplement gummies because they were mislabeled. According to a Nordic Naturals representative, the gummies contain sugar and fumaric acid, which are not listed on the label.
Recalls & Warnings
August 13, 2020
FDA Warns Three More Companies Selling Unapproved COVID-19 Tests
Between July 23 and 24, the FDA issued warning letters to three companies for marketing unapproved, adulterated or misbranded antibody tests for coronavirus (COVID-19).
Recalls & Warnings
May 09, 2020
FTC Warns 45 More Companies for Coronavirus Claims
On May 7, 2020, the FTC announced that it sent warning letters to 45 companies for selling products such as herbal products, immune system boosters, and vitamin C with unsupported claims that they can treat coronavirus (COVID-19).
Recalls & Warnings
March 04, 2022
FTC, FDA, and DOJ Take Joint Action Against Herbal Tea Companies for COVID Claims
On March 3, 2022, the FDA, FTC, and DOJ sued a New York-based marketer of herbal tea, in attempts to permanently block deceptive ads that claim Earth Tea is clinically proven to treat, cure, and prevent COVID-19.
Recalls & Warnings
March 09, 2023
"Dr. Rima Recommends" Nano Silver Recalled
On March 7, 2023, Natural Solutions Foundation issued a recall of Dr. Rima Recommends Nano Silver 10ppm Dietary Supplement, which was promoted to prevent and/or treat COVID-19.
Recalls & Warnings
July 14, 2020
Sellers of Miracle Mineral Solution Criminally Charged With Making Coronavirus Claims
On July 8, 2020, federal prosecutors charged the sellers of Miracle Mineral Solution, a toxic bleach solution marketed as a cure for coronavirus (COVID-19), with conspiracy to defraud the United States, conspiracy to violate the Federal Food, Drug and Cosmetic Act, and criminal contempt.
Recalls & Warnings
December 29, 2021
Natural Solutions Foundation Banned From Selling "Nano Silver" as COVID Treatment
On December 28, 2021, the United States Department of Justice announced that Natural Solutions Foundation has agreed to stop selling its nano silver products, in order to settle a suit brought against the company by the FDA for claiming the product could that the company claimed could prevent, ...
Recalls & Warnings
November 24, 2021
FDA Warns Sanitizer Corporation For Manufacturing and Misbranding Violations
On September 24, 2021, the FDA issued a warning letter to Chameleon Beverage Co. Inc.
Recalls & Warnings
July 25, 2024
West Coast Laboratories Warned for Manufacturing, Labeling Violations
On March 12, 2024, the FDA issued a Warning Letter to West Coast Laboratories, Inc.
Recalls & Warnings
March 13, 2025
FTC Sends Refund Checks to Consumers of Pure Green Coffee Supplement
On March 6, 2025, the Federal Trade Commission (FTC) announced it is mailing 39,977 checks totaling more than $905,000 to consumers who purchased Pure Green Coffee, to settle charges the product was promoted with deceptive health claims and marketing practices.
Recalls & Warnings
January 06, 2025
ZOE Prebiotic Blend Recalled Due to Metal Pieces, Stones
On December 4, 2024, Zoe US, Inc. recalled 142 units of ZOE Daily 30+ Prebiotic blend because it may contain foreign objects such as small metal pieces and/or stones.
Recalls & Warnings
October 06, 2005
Deceptive Marketing of “Supreme Greens" -- Settlement with FTC
On October 6, 2005, the Federal Trade Commission (FTC) announced that three individuals and two companies have settled FTC charges over their roles in the deceptive marketing of Supreme Greens, an herbal supplement.
Recalls & Warnings
March 03, 2012
FDA Warns Vitaganic of Manufacturing Violations
On February 8, 2012, the U.S. FDA sent a Warning Letter to Vitaganic, Inc. regarding violations of Current Good Manufacturing Practic (CGMP) regulations for dietary supplements found during an inspection of it manufacturing facility in Sunnyvale, California in October 2011.
Recalls & Warnings
April 28, 2020
Ten Multi-Level Marketing Companies Warned for Coronavirus and Deceptive Earnings Claims
On April 24, 2020, the FTC announced that it sent warning letters to ten multi-level marketing companies for selling products such as essential oils and immune system boosters with unsupported claims that they can treat coronavirus (COVID-19) and/or for misrespresenting potential earnings people ...
Recalls & Warnings
April 26, 2021
Family Indicted for Selling Dangerous "Bleach" Miracle Mineral Solution As "COVID" Cure
Four Florida men have been indicted by a Miami federal grand jury on charges of fraudulently marketing and selling the toxic bleach solution Miracle Mineral Solution (MMS) as a cure for COVID-19 and other serious medical conditions, such as cancer, Alzheimer's, diabetes, autism, malaria, ...
Recalls & Warnings
July 31, 2020
FTC Sues Golden Sunrise for Marketing Deceptive $23,000 Coronavirus Cure
On July 31, 2020, the Federal Trade Commission (FTC) charged Golden Sunrise Nutraceutical, Inc., a California-based company, with deceptively advertising a $23,000 treatment plan as a scientifically proven way to treat COVID-19.
Recalls & Warnings
July 26, 2023
Gadget Island, Inc. Male Enhancement Supplements Found to Contain Prescription Drugs
On July 21, 2023, the FDA issued a warning letter to Gadget Island, Inc.
Recalls & Warnings
April 01, 2004
Aloe Producer Recalls Product Due to Toxic Levels of Vitamin D
On March 26.2004, the Food and Drug Administration (FDA) announced that Aloe Commodities International, Inc., Carrollton, Texas, is recalling 1600 bottles of Solutions IE Ageless Formula II, Lot numbers P2207 and P2221 because they contain a significantly higher-than-labeled level of vitamin D3.
Recalls & Warnings
January 24, 2003
FTC Challenges Weight-loss Claims for Slim Down Solution
The Federal Trade Commission today charged Slim Down Solution, LLC, Maderia Management, Inc., and several related companies and individuals with using false and unsubstantiated claims in the marketing and advertising of "Slim Down Solution" - a purported weight-loss product.
Recalls & Warnings
August 13, 2020
FDA Warns Seller of Chaga for Coronavirus Claims
On August 6, 2020, the FDA issued a warning letter to Canadian Chaga for selling 124 Chaga Capsules, Chaga Tea, and Canadian Chaga Tincture with unsupported claims that they can treat coronavirus (COVID-19).
Recalls & Warnings
January 03, 2024
FDA Warns Amazon for Selling Men’s Supplements Containing Prescription Drugs
On December 20, 2023, the FDA issued a Warning Letter to Amazon.com, Inc.
Recalls & Warnings
September 06, 2023
WEFUN Capsules Recalled Due to Undeclared Sildenafil
On August 25, 2023, WEFUN Inc. issued a recall of 300 boxes of WEFUN Capsules after FDA analysis found the product to contain undeclared sildenafil, a prescription medication.
Recalls & Warnings
July 19, 2023
FDA Warns Seller of Sleepy Time Products and Honey Herbal Syrups for Manufacturing Violations
On June 28, 2023, the FDA issued a warning letter to Eden’s Answers, Inc. after an inspection of the company’s facility found products to be adulterated because they were prepared, packed, or held under conditions that do not meet Current Good Manufacturing Practices (CGMP).
Recalls & Warnings
November 09, 2023
InnoMark Warned for Manufacturing & Misbranding Violations
On September 1, 2023, the FDA issued a Warning Letter to InnoMark, Inc.
Recalls & Warnings
June 19, 2023
A&Z Pharmaceutical Warned for Promoting Calcium & Vitamin D for Hair Loss, Osteoporosis & Cancer
On June 1, 2023, the FDA issued a warning letter to A&Z Pharmaceutical, Inc. following inspection of the company’s website and social media, which found statements about the company’s Chewable Calcium 600MG with Vitamin D for Kids in Orange Flavor products to be drug claims.
Recalls & Warnings
July 07, 2020
Seller of Homeopathic Products Warned for Manufacturing Violations
On June 19, 2020, the FDA issued a warning letter to Washington Homeopathic Products, Inc.
Recalls & Warnings
September 08, 2020
Seller of "Diabetes Supplement" & More Warned for Drug Claims
On August 25, 2020, the FDA issued a warning letter to Nutritional Supplements Corporation Inc following a review of the company's website, which found statements made about some of the company's products, including Vitadone, Diabrex, and Viadevita to be drug claims.
Recalls & Warnings
September 08, 2020
FDA Warns Seller of Multivitamin for Manufacturing Violations
On August 14, 2020, the FDA issued a warning letter to Revival Products, Inc.
Recalls & Warnings
November 10, 2020
FDA Warns Seller of Digestive Enzymes, Nattokinase, and More
On October 27, 2020, the FDA issued a warning letter to World Nutrition, Inc.
Recalls & Warnings
June 04, 2004
FTC Charges Marketers of Two Supplements with False Claims to Cure Range of Diseases
On June 3, 2004 the Federal Trade Commission (FTC) charged marketers of two dietary supplements with falsely claiming that their products can prevent and cure cancer and other diseases. According to the FTC’s complaint, Boston-area marketers Direct Marketing Concepts, Inc. (DMC), ITV Direct, Inc.
Recalls & Warnings
May 23, 2013
Maker of Liver Detox and Insulin Supplements Warned For Drug Claims
On April 24, 2013, the FDA issued a warning letter to Glucorell, Inc.
Recalls & Warnings
August 12, 2019
Miracle Mineral Solution Is Dangerous, Warns FDA
On August 12, 2019, the FDA warned consumers not to buy or use Miracle Mineral Solution or other "Miracle" or "Master" solution products containing sodium chlorite because they can dangerous and even life-threatening reactions.
Recalls & Warnings
August 13, 2020
FDA Warns Soluciones Cosmeticas for Hand Sanitizers That May Contain Toxic Ingredient
On August 4, 2020, the FDA issued a warning letter to Soluciones Cosmeticas, SA de CV which after FDA testing found samples of the company's BERSIH Antiseptic Alcohol 70% Topical Solution Hand Sanitizer and BERSIH HAND SANITIZER GEL Fragrance Free to contain methanol, a toxic ...
Recalls & Warnings
July 06, 2022
Two Companies Banned From Selling Supplements to Treat Heart Disease, Neuropathy
On June 30, 2022, the Federal Trade Commission (FTC) finalized an administrative complaint order against two Texas-based companies, Health Research Laboratories, LLC and Whole Body Supplements, LLC, for making unverified claims that their products can prevent or treat disease.
Recalls & Warnings
July 14, 2022
FDA Warns Seller of Unauthorized COVID-19 Tests
On June 30, 2022, the FDA issued a warning letter to W.H.P.M, Inc. for distributing COVID antigen tests without approval, clearance, or authorization from the FDA while claiming to mitigate, prevent, treat, diagnose, or cure COVID-19 in people.
Recalls & Warnings
June 13, 2022
Homeopathic Nasal Spray SnoreStop Recalled Due to Microbial Contamination
On June 9, 2022, Green Pharmaceuticals Inc issued a voluntary nationwide recall of one lot of its SnoreStop NasoSpray, a homeopathic nasal spray promoted to reduce snoring, due to the presence of Providencia rettgeri, a bacterium that can cause severe illness in some people.
Recalls & Warnings
July 25, 2022
Nature’s Sunshine Protein Powder Recalled
On July 22, 2022, Nature’s Sunshine Products Inc. issued a recall of two AIVIA Whey Protein Powder Herbs meal replacement products due to undeclared milk.
Recalls & Warnings
January 19, 2023
Male Sexual Enhancement Supplement Adam’s Secret Found to Contain Prescription Medication
On January 10, 2023, the FDA issued a warning letter to HIS Enterprise Inc dba Adam’s Secret USA, LLC after laboratory analysis found Adam’s Secret Extra Strength 3000 Platinum, Adam’s Secret Extra Strength Blue, Adam’s Secret Extra Strength Purple, Adam's Secret ...
Recalls & Warnings
January 26, 2023
FDA Warns Seller of Canary Seed Omega-3
On November 16, 2022, the FDA issued a warning letter to Evimeria El Aztecano, Inc. following review of the company’s Yerbas Finas Leche de Alpiste con Guanabana y Omega-3 product labeling, which found the product contained less magnesium and potassium than claimed on the label.
Recalls & Warnings
December 21, 2019
Supplement Company Continues to Make False Claims About Its Products, Says FTC
The FTC has filed a motion of contempt against two supplement companies that, according to the motion, have continued to promote their products with false claims despite being barred from doing so by a previous court order.
Recalls & Warnings
July 28, 2020
Four More Companies Recall Hand Sanitizers
Between July 9 and 27, 2020, four more companies recalled hand sanitizers because they may contain methanol, which is toxic when absorbed through the skin or ingested:
Recalls & Warnings
March 09, 2021
FDA Warns Sellers of Three More Hand Sanitizers, One with Toxic Ingredient
On March 1, 2021, the FDA issued warning letters to three sellers of hand sanitizers because laboratory tests showed that their products do not contain the amount of ethanol as stated on their labels. The products are MEDICALLY MINDED Hand Sanitizer Gel from Plasticos Las Palmas, S.A. de C.
Recalls & Warnings
July 06, 2020
FDA Warns Five More Hand Sanitizers May Contain Toxic Ingredient
Update: (7/14/20) The FDA has warned consumers not to use forty-six more hand sanitizers that may contain methanol.
Recalls & Warnings
November 15, 2009
Nationwide Recall of a Weight Loss Supplement Found to Contain Undeclared Drug Ingredients
On November 12, 2009, The U.S. Food and Drug Administration (FDA) announced that it informed GMP Herbal Products, Inc. that its product "Pai You Guo," a weight loss dietary supplement, contains undeclared drug ingredients.
Recalls & Warnings
April 16, 2019
DMHA and Phenibut Are Not Permitted in Dietary Supplements, Warns FDA
On April 16, 2019, the FDA announced it has issued 11 warning letters to companies whose dietary supplement products contain the drugs DMHA or phenibut, and therefore are in violation of the law.
Recalls & Warnings
March 12, 2016
FDA Finds Problems at 58% of Supplement Manufacturing Sites in U.S. and Abroad
ConsumerLab.com has obtained results of the FDA's inspections in Fiscal Year 2015 (ending September 30) of 483 dietary supplement manufacturing facilities, showing that most -- 58.2% -- received letters indicating noncompliance with current Good Manufacturing Practices (cGMPs).
Recalls & Warnings
November 28, 2012
Maker of Pycnogenol, Memory and Immune Supplements Warned For Manufacturing Violations and Misbranding
On November 15, 2012, the FDA issued a warning letter to contract manufacturer Health Technology, Inc.
Recalls & Warnings
March 13, 2020
State of Missouri Sues Jim Bakker Show for Selling False Coronavirus Cure
On March 12, 2020, the Missouri Attourney General announced that it sued the Jim Bakker Show for selling a fake coronavirus (COVID-19) cure called Silver Solution.
Recalls & Warnings
February 09, 2022
FDA Warns Seller of Colloidal Silver Eye Drops, Copper Products & More
On February 1, 2022, the FDA issued a warning letter to New Earth Healing Essentials, LLC d/b/a 5D Full Disclosure following a review of the company’s website, which found statements made about some of the company's products, including Plasma Colloidal Silver Eyedrops, Gaia’s ...
Recalls & Warnings
April 27, 2022
FDA Warns Manufacturer of Topical Antiseptic Products for COVID Claims
On April 19th, 2022, the FDA issued a warning letter to Kleenhanz, LLC following a review of the company’s website and social media which found statements about the company’s Kleenhanz Towelettes topical antiseptic products to be drug claims.
Recalls & Warnings
April 25, 2022
Some OTC Skin Lighteners Contain Potentially Harmful Ingredient, Warns FDA
On April 19th, 2022, the FDA issued warnings to 12 companies for selling over-the-counter (OTC) skin-lightening products containing hydroquinone.
Recalls & Warnings
May 29, 2021
FDA, FTC Warns Five Sellers of "Infertility" Supplements
The FDA and FTC (Federal Trade Commission) sent warning letters to the following five companies in May for illegally selling dietary supplements promoted with claims to treat infertility and other reproductive health issues:
Recalls & Warnings
December 26, 2010
Four Probiotic Products in Canada May Pose Serious Health Risks to Those with Milk or Soy Allergies
On December 24, 2010, Health Canada (the Canadian health ministry) advised consumers with milk or soy allergies that four probiotic natural health products are being voluntarily recalled from the market because they are labelled as not containing dairy (milk) and/or soy but may contain trace ...
Recalls & Warnings
January 09, 2009
FDA Expands Warning to Consumers About Tainted Weight Loss Pills
On January 8, 2009, the U.S. FDA expanded its nationwide alert to consumers about tainted weight loss pills that contain undeclared, active pharmaceutical ingredients. On December 22, 2008, FDA warned consumers not to purchase or consume 28 different products marketed for weight loss.
Recalls & Warnings
November 24, 2018
Dangerous Drug In Supplements for Pain and Anxiety
On November 20, 2018, the FDA announced it has issued warning letters to two companies for the illegal marketing of products labeled as dietary supplements that contain the opioid-like drug tianeptine.
Recalls & Warnings
December 26, 2017
Seller of BrainAlert Warned for Manufacturing Violations, Drug Claims
On December 14, 2017, the FDA issued a warning letter to BrainAlert, LLC.
Recalls & Warnings
April 11, 2020
FDA Warns Sellers of CBD, Colloidal Silver & Natural Remedies Promoted to Treat Coronavirus
Between April 7 and April 9, 2020, the FDA issued warning letters to five companies for selling products such as CBD, colloidal silver, and natural treatments with unsupported claims that they can treat coronavirus (COVID-19).
Recalls & Warnings
March 02, 2021
FDA Warns Makers of Four Hand Sanitizers, Two With Toxic Ingredient
Between February 18 and 25, 2021, the FDA issued warning letters to four sellers of hand sanitizers because laboratory tests showed that their products do not contain the amount of ethanol as stated on their labels.
Recalls & Warnings
April 17, 2021
NS NY Distributor Inc Male Enhancement Products Recalled Due to Undeclared Drugs
On April 8, 2021, NS NY Distributor Inc issued a recall of all lots of Premium Orgazen 7000 and Ginseng Power 5000 male enhancement capsules because FDA analysis found them to contain sildenafil and/or tadalafil.
Recalls & Warnings
January 10, 2012
Acai Berry Pill Marketers to Pay $1.5 Million to Settle FTC Charges
On January 9, 2012, the U.S. Federal Trade Commission (FTC) announced that an operation that marketed acai berry supplements, "colon cleansers," and other products using allegedly fraudulent free trial offers and phony endorsements from Oprah Winfrey and Rachael Ray will pay $1.
Recalls & Warnings
August 02, 2012
FDA Warns Two Supplement Makers of Manufacturing Violations
On July 19, 2012, the FDA issued a warning letter to H & L Jerch Sales, Inc. for failing to maintain written quality control procedures, which constitutes a violation of current Good Manufacturing Practices (cGMP) and caused the company’s products to be declared adulterated.
Recalls & Warnings
September 02, 2016
Maker of Prescription Multi to Pay $15.5 Million to Settle Lawsuit Over Ingredient Claim
Endo Health Solutions Inc. has reportedly agreed to pay $15.5 million to settle a class action lawsuit which alleged its Qualitest Multi-Vitamin with Fluoride Chewable Tablets contained just 44% of the fluoride claimed on the label.
Recalls & Warnings
October 30, 2014
Seller of Vitamin C, Iron and Detox Supplements Warned for Violations, Drug Claims
On October 16, 2014, the FDA issued a warning letter to Vitalab Co., Inc. (which manufactures and labels supplements for three distributors: V.E. Irons, Inc., Springreen Products, Inc., and Sonne's Organic Foods, Inc.
Recalls & Warnings
October 18, 2012
Natural Product Manufacturer Warned For GMP Violations, Misbranding, Unapproved Devices and Drug Claims
On August 28, 2012, the FDA issued a warning letter to Creation's Garden Natural Products, Inc. following an inspection of the company's manufacturing facility, which found violations of Current Good Manufacturing Practices (CGMPs) for dietary supplements.
Recalls & Warnings
February 11, 2012
Herbal Sprays for Colds and Cold Sores Recalled
On February 10, 2012 the U.S. FDA posted a notice announcing the recall of all "Koff & Kold" and "Kold Sore" sprays by Wholistic Herbs Inc. The products were distributed throughout the United States.
Recalls & Warnings
August 31, 2009
Court Orders Marketers of Supreme Greens and Coral Calcium to Pay Nearly $70 Million for Consumer Refunds
On August 27, 2009, a federal district court ordered the marketers of two dietary supplements – "Supreme Greens" and "Coral Calcium" – who claimed the products would cure ailments ranging from cancer and Parkinson’s disease to heart disease and autoimmune diseases to pay nearly $70 million for ...
Recalls & Warnings
July 13, 2012
Alistrol Health Inc. Warned Over Health Claims on Products
On June 26, 2012, the FDA issued a warning letter to Alistrol Health Inc. for making product statements on the company's websites that constitute drug claims.
Recalls & Warnings
March 31, 2018
Supplement Manufacturer Shut Down for Manufacturing Violations
On March 29, 2018, the FDA announced that U.S. District Court for the Eastern District of New York has entered a consent decree of permanent injunction with Riddhi USA, Inc. and Mohd M. Alam, president and owner of Riddhi USA, Inc., for selling adulterated and misbranded dietary supplements.
Recalls & Warnings
August 20, 2019
Seller of Liposomal Curcumin, Vitamin C & Melatonin Warned for Manufacturing Violations
On August 6, 2019, the FDA issued a warning letter to Let's Talk Health, Inc.
Recalls & Warnings
January 24, 2020
Fat-Burning, Energy Supplement Linked to Heart Trouble
A 33-year-old woman in Australia developed cardiac ischemia (a lack of blood flow to the heart) after taking the "fat burning" supplement Alpha Lean-7, according to a recent report in the Journal of Sports Sciences.
Recalls & Warnings
November 27, 2020
Harmonic Nature Hand Sanitizer Potentially Toxic
On November 16, 2020, the FDA issued a warning letter to Harmonic Nature S. de R.L. MI. because laboratory tests showed that their product Alcohol Antiseptic 75% Topical Solution Hand Sanitizer contains 1-propanol, which is toxic when ingested.
Recalls & Warnings
August 18, 2020
Colloidal Silver Seller Warned for Coronavirus Claims
On August 14, 2020, the FDA issued a warning letter to SilveryGuy for selling colloidal silver products with unsupported claims that they can treat coronavirus (COVID-19).
Recalls & Warnings
August 18, 2020
Seller of Immune Shot Criminally Charged With Making Coronavirus Claims
On August 10, 2020, prosecutors in Georgia charged Matthew Ryncarz and his company Fusion Health and Vitality, LLC d/b/a/ Pharm Origins with selling the misbranded product Immune Shot.
Recalls & Warnings
July 10, 2020
Seller of Hand Sanitizer "Alternative" Warned for Coronavirus Claims
On July 7, 2020, the FDA issued a warning letter to Ionogen, LLC for selling products with unsupported claims that they can treat coronavirus (COVID-19).
Recalls & Warnings
July 17, 2020
Sellers of Immune Boosters, Vitamin C Warned for Coronavirus Claims
The FDA recently issued warning letters to two companies for selling products such as immune boosters, vitamin C, and zinc with unsupported claims that they can treat COVID-19 (use the links below to read the full warning letter):
Recalls & Warnings
August 15, 2020
FTC Warns 20 More Companies for Coronavirus Claims
On August 14, 2020, the FTC announced that it sent warning letters to 20 companies for selling products such as vitamin C, hydrochloroquine, omega 3, and melatonin with unsupported claims that they can treat coronavirus (COVID-19).
Recalls & Warnings
May 19, 2020
FTC Sends Refund Checks to Consumers of Unproven Weight Loss and Sexual Enhancement Supplements
On May 19, 2020, the FTC announced it is mailing 143,636 checks totaling more than $8,500,000 to consumers who purchased deceptively marketed supplements.
Recalls & Warnings
March 05, 2021
FTC Takes Further Action Against Deceptive CBD Claims
On March 5, 2021, the Federal Trade Commission (FTC) announced that it has approved final administrative consent orders against six companies for selling CBD products with unsupported and deceptive health claims that they can treat a variety of conditions, including cancer, heart disease, ...
Recalls & Warnings
April 04, 2012
Maker of Protein Drinks and Supplements Warned of Manufacturing Violations
The FDA published a Warning Letter to Protica, Inc., a maker of protein drinks and supplements, regarding violations of manufacturing regulations discovered during inspection of its facility in Whitehall, PA. Affected products include the foods Amped Up-2oz, Fireball-2oz, Healthy Shot-2.
Recalls & Warnings
February 28, 2013
Maker of Liquid Omega-3 Supplements Warned For Manufacturing Violations
On February 20, 2013, the FDA issued a warning letter to Capco Custom Packaging Inc.
Recalls & Warnings
October 29, 2016
Miracle Mineral Solution Danger
The husband of a woman who died hours after drinking the liquid supplement Miracle Mineral Solution in 2009 believes the supplement caused her death, according a recent ABC News article and feature segment on ABC's 20/20 in October 2016.
Recalls & Warnings
June 19, 2018
FDA Warns Seller of Weight Control Patches
On June 6, 2018, the FDA issued a warning letter to Health Management Group, Inc.
Recalls & Warnings
February 04, 2017
Five Weight Loss Supplements Found to Contain Drugs
The FDA warned consumers not to buy or use the following weight loss supplements because they were found to contain undeclared drugs:
Recalls & Warnings
June 20, 2014
FDA Warns Some Bee Pollen Products for Weight Loss Are Dangerous
On June 19, 2014, the FDA warned consumers that some weight loss products containing bee pollen have been found to contain hidden drugs.
Recalls & Warnings
January 09, 2014
Marketers of "Genetically Customized" Supplements Settle FTC Charges of Deceptive Health Claims
Two marketers of "genetically customized" nutritional supplements have agreed to a settlement with the Federal Trade Commission (FTC) over charges that the companies made deceptive advertising claims and did not adequately protect customers' medical and financial information.
Recalls & Warnings
October 24, 2003
Court Closes the Doors on Company That Sold Weight Loss Supplement
On October 24, 2003, the Federal Trade Commission (FTC)announced that Mark Nutritionals, Inc. and Edward D’Alessandro, Jr. have agreed to settle federal charges that they used false and unsubstantiated claims to sell their weight-loss product.
Recalls & Warnings
February 27, 2009
$4 Million Settlement by Supplement Maker for False Claims
On February 26, 2009, the Texas Attorney General announced that an agreement was reached with Mannatech Inc. and its former CEO, Samuel L. Caster, both of which had been charged with orchestrating an unlawful marketing scheme that exaggerated their products’ health benefits.
Recalls & Warnings
August 09, 2010
FDA Warns Against "Miracle" Mineral Supplement
On July 30, 2010, The U.S. FDA warned consumers not to consume or use Miracle Mineral Solution, an oral liquid solution also known as "Miracle Mineral Supplement" or "MMS." The product, when used as directed, produces an industrial bleach that can cause serious harm to health.
Recalls & Warnings
September 26, 2014
FDA Warns Seller of Supplements and Chocolate Promoted to Treat Ebola
On September 23, 2014, the FDA issued a warning letter to Natural Solutions Foundation, following a review of the company's websites which found statements made about Silver Sol Nano Silver (also called The Silver Solution) and CBD Organic Dark Chocolate Bars (also called High Potency CBD Hemp Oil) ...
Recalls & Warnings
March 31, 2005
FDA Warns Marketer of "Vitamin O" Product to Cease Unsubstantiated Claims
The U.S. Food and Drug Adminstration (FDA) has sent a Warning Letter (dated February 8, 2005) to Donald L. Smyth, President, R-Garden Inc., and Rose Creek Health Products, Inc. warning that its "Vitamin O" product was, among other things, being marketed with unsubstantiated health claims.
Recalls & Warnings
January 17, 2006
FDA Warns Consumers about Brazilian Diet Pills Found to Contain Active Drug Ingredients
On January 13,2006, the U.S. Food and Drug Administration (FDA) warned consumers not to use two unapproved drug products that are being marketed as dietary supplements for weight loss.
Recalls & Warnings
November 27, 2011
FDA Seeks Permanent Injunction Against Dietary Supplement Maker -- 400+ Products Affected
On November 23, 2011, the U.S. FDA took legal action against a dietary supplement maker and owner for substituting ingredients and products without noting the changes on the final product labels. The permanent injunction, filed on behalf of the FDA by the U.S.
Recalls & Warnings
October 25, 2013
Supplement Company Warned For Numerous Manufacturing Violations
On August 2, 2013, the FDA issued a warning letter to DNE Nutraceuticals, Inc., following a facility inspection which found the company's products to be adulterated because they were packed, or held under conditions that violate Current Good Manufacturing Practices for dietary supplements.
Recalls & Warnings
March 14, 2012
Manufacturing Violations by Maker of Lecithin, Soy Supplements
The FDA issued a Warning Letter (dated March 7, 2012) to Modern Products, Inc. regarding manufacturing violations in the production of the supplements Fearn Lecithin Granules, Fearn Liquid Lecithin, Fearn 100% Soy Protein Isolate, and Gayelord Hauser Brewers Yeast.
Recalls & Warnings
February 06, 2013
Ingredient Supplier Warned For Meal Replacement Product Misbranding and Adulteration
On October 4, 2012, the FDA issued a warning letter to dietary supplement ingredient supplier Raw Deal, Inc.
Recalls & Warnings
March 08, 2016
Maker of Calcium and Vitamin C Supplements Warned for Manufacturing Violations
On September 17, 2015, the FDA issued a warning letter to Raphah, Inc.
Recalls & Warnings
September 25, 2018
Seller of B Vitamins, Multis, Glucosamine & More Warned for Manufacturing Violations
On August 31, 2018, the FDA issued a warning letter to Independent Nutrition Inc., following a facility inspection which the company's products, including B-50 Complete, Multi-Vitamin & Mineral Complex (a.k.a.
Recalls & Warnings
September 12, 2018
FDA Urges Consumers to Avoid Kratom
On September 11, 2018, FDA Commissioner Scott Gottlieb, M.D., urged consumers not to use products containing the kratom, an herb that is often promoted for pain relief and for relieving symptoms of opioid withdrawal.
Recalls & Warnings
January 04, 2007
Sellers of Popular Weight Loss Supplements Pay $25 Million Over FTC Allegations of Deceptive Advertising
On January 4, 2007, the Federal Trade Commission (FTC) announced that it had filed complaints in four separate cases alleging that weight-loss and weight-control claims were not supported by competent and reliable scientific evidence.
Recalls & Warnings
May 24, 2013
Seller of Liquid Minerals, Joint Care and Herbal Supplements Warned For Manufacturing Violations, Drug Claims
On April 8, 2013, the FDA issued a warning letter to Body Systems, Inc.
Recalls & Warnings
July 05, 2013
Seller of Vision Supplement Warned For Drug Claims
On June 25, 2013, the FDA issued a warning letter to Nutrient Synergy, Inc., following a review of the company's website which found statements made about the dietary supplement Nepretin to be drug claims.
Recalls & Warnings
April 18, 2013
Cardio, Energy and Sexual Enhancement Supplement Distributor Warned For Manufacturing Violations and Drug Claims
On December 21, 2012, the FDA issued a warning letter to ForMor Inc, dba ForMor International, following a facility inspection which found the company's Cardio Cocktail and Argenix dietary supplements to be adulterated because because they were prepared, packed, or held under conditions that do ...
Recalls & Warnings
February 17, 2018
FDA Warns Seller Reishi Mushroom Supplements
On February 7, 2018, the FDA issued a warning letter to Reishi D. International, Inc., following a facility inspection which found the company's Reishi D.
Recalls & Warnings
May 19, 2018
Homeopathic Teething Drops, Nausea Drops, Silver-Zinc Throat Spray & More Recalled
On May 18, 2018, MBI Distributing, Inc.
Recalls & Warnings
February 07, 2017
Seller of Chinese Herbal Products for Prostate, Menopause and More Warned for Manufacturing Violations
On January 23, 2017, the FDA issued a warning letter to Herbal Sciences International, Inc.
Recalls & Warnings
July 23, 2016
Maker of "Super Food" Warned for Manufacturing Violations
On July 12, 2016, the FDA issued a warning letter to TerraVare, Inc.
Recalls & Warnings
July 18, 2017
FDA Warns Seller of "Quick Slim with pure Hoodia Gardonii" and "Diabalance Herbal Blood Sugar Balance"
On July 11, 2017, the FDA issued a warning letter to Black Seed Herb, Inc.
Recalls & Warnings
September 26, 2015
Seller of Muscle Supplements and More Warned for Manufacturing Violations
On August 14, 2015, the FDA issued a warning letter to Chaotic Labz, Inc.
Recalls & Warnings
January 31, 2015
Maker of Soy and Zinc Warned for Manufacturing Violations
On December 18, 2014, the FDA issued a warning letter to PreMark Health Science, Inc.
Recalls & Warnings
April 29, 2014
Seller of B Vitamins, CoQ10, Amino Acids and Other Supplements Warned for Manufacturing Violations and Drug Claims
On April 1, 2014, the FDA issued a warning letter to Bio-Recovery, Inc.
Recalls & Warnings
February 19, 2014
Seller of Sexual Enhancement Supplements Warned For Manufacturing Violations
On February 11, 2014, the FDA issued a warning letter to Maximus Niterider International Group, Inc.
Recalls & Warnings
December 22, 2008
FDA Warns Consumers About Tainted Weight Loss Pills
On December 22, 2008 the U.S. FDA alerted consumers not to purchase or consume any of more than 25 different products marketed for weight loss because they contain undeclared, active pharmaceutical ingredients that may put consumers’ health at risk.
Recalls & Warnings
October 06, 2004
False Claims Made by Marketer of Cortisol-related Weight-Loss Supplements According to FTC
On October 6, 2004, the Federal Trade Commission (FTC)charged marketers of two dietary supplements with claiming, falsely and without substantiation, that their products can cause weight loss and reduce the risk of, or prevent, serious health conditions.
Recalls & Warnings
November 21, 2014
Seller of Silver Supplement Warned for Claiming to Treat Ebola and Other Viruses
On November 19, 2014, the FDA issued a warning letter to LifeSilver.com, following a review of the website, which found statements made about Silver sol to be drug claims.
Recalls & Warnings
March 28, 2011
Dangerously High Levels of Vitamins A and D in Product Prompt FDA Warning
On March 28, the U.S. FDA warned consumers to stop using Soladek, a vitamin-solution product marketed by Indo Pharma, S.A., of the Dominican Republic, because the product may contain dangerously high levels of vitamins A and D.
Recalls & Warnings
May 01, 2009
FDA Warns Consumers to Stop Using Hydroxycut -- Product Being Tested by ConsumerLab.com
On May 1, 2009, the U.S. Food and Drug Administration warned consumers to immediately stop using Hydroxycut products by Iovate Health Sciences Inc., of Oakville, Ontario and distributed by Iovate Health Sciences USA Inc. of Blasdell, N.Y.
Recalls & Warnings
July 15, 2010
Supplement Company Pays $5.5 Million to Settle False Advertising Claims
On July 14, 2010, the Federal Trade Commission (FTC), as part of its ongoing efforts to stop bogus health claims, announced that it is requiring a major marketer of dietary supplements to pay $5.
Recalls & Warnings
March 20, 2020
Seller of Rejuvenation Pills Settles Charges of Making False Claims
Health Center, Inc. has agreed to halt their allegedly deceptive advertising claims about three "cure-all" health and wellness products that targeted older consumers nationwide after the Federal Trade Commission (FTC) filed a complaint.
Recalls & Warnings
April 21, 2020
U.S. Attorney's Office Bars Chiropractor from Selling Fake Coronavirus Cures
On April 17, 2020, the United States Attorney's Office for the Northern District of Texas announced that it has obtained a temporary restraining order preventing a chiropractor from promoting fake treatments for coronavirus (COVID-19). Dr. Ray L.
Recalls & Warnings
December 27, 2019
Federal Court Shuts Down Three Supplement Companies With Serious Manufacturing Violations
Update: (1/23/20) ABH has issued a recall of all its products, which are sold under various brand names by over 800 distributors and retailers. More details are available in CL's post about this recall.
Recalls & Warnings
November 26, 2019
FDA Warns Companies Selling CBD Products as Dietary Supplements
On November 25, 2019, the FDA issued warning letters to 15 companies for selling products containing CBD (cannabidiol) labeled and marketed as dietary supplements, and/or for making drug claims about these products.
Recalls & Warnings
August 30, 2012
Supplement Company Warned For Medical Claims and Misbranding Of Omega-3, CoQ10, Noni Juice And More
On July 12, 2012, the FDA issued a warning letter to Alfa Vitamins Laboratories, Inc.
Recalls & Warnings
October 21, 2014
Seller of Weight Loss Supplements Warned for Manufacturing Violations and Drug Claims
On October 7, 2014, the FDA issued a warning letter to YoungYou International, Inc.
Recalls & Warnings
May 21, 2014
Seller of Gingko and Milk Thistle Warned for Manufacturing Violations and Drug Claims
On April 4, 2014, the FDA issued a warning letter to Xtra Life Natural Systems, Inc.
Recalls & Warnings
September 09, 2014
Maker of Popular Green Coffee Bean Extract Settles FTC Charges of Unsupported Weight Loss Claims
Applied Food Sciences, Inc., maker of green coffee bean extract GCA, has agreed to pay $3.5 million to settle FTC charges that weight loss claims made about the extract were not supported by scientific evidence.
Recalls & Warnings
July 26, 2014
Marketers of Nopal Cactus Drink Settle FTC Charges of Deceptive Claims
TriVita Inc., and marketers of the company's "prickly pear" fruit drink Nopalea have agreed to pay $3.5 million in consumer refunds to settle FTC charges they made deceptive claims that the drink treats health problems ranging from skin conditions to joint pain and respiratory problems.
Recalls & Warnings
January 27, 2015
Seller of Aloe, Sexual Enhancement Supplements and More Warned for Manufacturing Violations, Drug Claims
On January 8, 2015, the FDA issued a warning letter to Aloe Man, Inc., following a facility inspection which found the company's products, including The Aloe Man's Super Bright, Dr. Johnson's Maximum Desire, Dr. Johnson's Body Healer, and Dr.
Recalls & Warnings
January 23, 2015
Maker of Vitamin Drinks Warned for Manufacturing Violations
On January 7, 2015, the FDA issued a warning letter to NYSW Beverage Brands, Inc.
Recalls & Warnings
January 17, 2015
Maker of Magnesium and Potassium Supplements Warned for Manufacturing Violations
On December 23, 2014, the FDA issued a warning letter to Nutri Spec, Inc.
Recalls & Warnings
July 28, 2015
Abnormal Heart Rhythms Linked with Unauthorized Natural Product, Health Canada Warns
On July 27, 2015, Health Canada (the Canadian equivalent of the U.S. FDA) announced that it has received a serious adverse reaction report of abnormal heart rhythms associated with the use of an unauthorized natural product in Canada called Remogen, which contains the drug ibogaine.
Recalls & Warnings
July 02, 2015
Maker of Arthritis Supplement Warned for Manufacturing Violations, Drug Claims
On June 17, 2015, the FDA issued a warning letter to Desert Stream, Inc.
Recalls & Warnings
May 23, 2015
Seller of Magnesium, Calcium, Silver and More Warned for Manufacturing Violations, Drug Claims
On May 8, 2015, the FDA issued a warning letter to Pick and Pay, Inc.
Recalls & Warnings
May 13, 2015
Amberen Weight Loss Claims Not Supported by Evidence, Says FTC
On May 12, 2015, the FTC announced it filed a complaint to stop Lunada Biomedical Inc. from advertising that its product Amberen is clinically proven to cause substantial weight loss in women over 40.
Recalls & Warnings
September 18, 2015
High Levels of Mercury and Lead Found in Ayurvedic Herbal Supplements
On September 17, 2015, the FDA announced that Shree Baidyanath Ayurved Bhawan Ayurvedic dietary supplements were found to contain high levels of lead and/or mercury, which can cause serious health problems.
Recalls & Warnings
February 06, 2016
Seller of Turmeric, Milk Thistle and More Warned for Manufacturing Violations, Drug Claims
On January 15, 2016, the FDA issued a warning letter to Terra Firma Botanicals, Inc.
Recalls & Warnings
February 03, 2016
Seller of Weight and Enhancement Supplements Warned For Drug Claims
On December 11, 2015, the FDA issued a warning letter to The One Minute Miracle, Inc., following a review of the company's website which found statements made about Miracle Diet 30 and Miracle Rock 48 to be drug claims.
Recalls & Warnings
January 20, 2016
Maker of Growth Hormone, Testosterone Booster Warned for Manufacturing Violations, Misbranding
On January 8, 2016, the FDA issued a warning letter to Nutraloid Labs Inc.
Recalls & Warnings
December 29, 2015
Maker of Vitamin K, Vitamin A & More Warned for Manufacturing Violations, Drug Claims
On December 10 2015, the FDA issued a warning letter to Dherbs Health Emporium, Inc.
Recalls & Warnings
June 27, 2017
Seller of Alpha Lipoic Acid, Cinnamon Supplements and More Warned For Manufacturing Violations
On May 1, 2017 the FDA issued a warning letter to Nature's Vision, Inc.
Recalls & Warnings
May 30, 2017
Maker of Collagen Supplement Warned for Manufacturing Violations
On July 17, 2014, the FDA issued a warning letter to Morhaim Pharmalab, Inc.
Recalls & Warnings
September 01, 2018
Homeopathic Remedies Contaminated With Bacteria
On August 29, 2018, Hellolife, Inc.
Recalls & Warnings
April 04, 2017
Seller of B Vitamins, Vitamin C, Potassium & More Warned for Manufacturing Violations
On March 24, 2017 the FDA issued a warning letter to The Sanapac Company, Inc.
Recalls & Warnings
March 15, 2016
FDA Warns Sellers of Weight and Workout Supplements Containing Acacia Rigidula
On March 7, 2016, the FDA issued warning letters to five sellers of supplements which were labeled as containing Acacia rigidula, an ingredient which is not permitted in dietary supplements.
Recalls & Warnings
August 31, 2016
Seller of Joint Supplement Warned for Manufacturing Violations, Drug Claims
On July 15, 2016, the FDA issued a warning letter to Vitalife Inc.
Recalls & Warnings
February 09, 2017
Federal Court Orders Dietary Supplement Distributor to Stop Selling Its Products
On February 9, 2017, the FDA announced that VivaCeuticals Inc, doing business as Regeneca Worldwide, has been ordered by a federal court to stop selling its products, which were found to contain unsafe ingredients including DMAA.
Recalls & Warnings
October 08, 2016
Seller of Cell Power and Super Silica Warned for Manufacturing Violations, Misbranding
On September 23, 2016, the FDA issued a warning letter to SSO, Inc.
Recalls & Warnings
September 14, 2016
Seller of Aloe, Prostate and Joint Supplements Warned for Manufacturing Violations
On April 8, 2016, the FDA issued a warning letter to Salud Natural Entrepreneurs, Inc.
Recalls & Warnings
May 15, 2018
Seller of Apple Cider Vinegar, Joint Supplements & More Warned for Drug Claims
On April 25, 2018, the FDA issued a warning letter to Baker's Best Health Products, Inc.
Recalls & Warnings
December 26, 2017
Seller of Fish Oil, Vitamin D, Whey Protein and More Warned for Manufacturing Violations
On December 19, 2017, the FDA issued a warning letter to Maine Natural Health, Inc.
Recalls & Warnings
December 23, 2017
Seller of Meal Replacement, Protein Drinks, Cranberry and More Warned for Manufacturing Violations
On July 6, 2017, the FDA issued a warning letter to Professional Botanicals, Inc.
Recalls & Warnings
October 13, 2017
Hi-Tech To Pay Over 40 Million to Settle FTC Charges of False Claims
On October 11, 2017, a U.S. District Judge ruled that Hi-Tech Pharmaceuticals Inc.
Recalls & Warnings
August 29, 2017
FDA Warns Seller of Supplements for Allergies, Joint Pain, Bone Health, and More
On August 16, 2017, the FDA issued a warning letter to Total Nutrition, Inc.
Recalls & Warnings
June 05, 2018
FDA Warns Seller of Colloidal Silver
On May 17, 2018, the FDA issued a warning letter to Silver Armor, Inc.
Recalls & Warnings
September 19, 2018
Seller of Curcumin, Garcinia, Glucosamine and More Warned for Manufacturing Violations
On September 7, 2018, the FDA issued a warning letter to Best Nutrition Products, Inc.
Recalls & Warnings
December 12, 2018
Seller of Protein Powder, Spirulina & Other Products Warned for Manufacturing Violations
On October 31, 2018, the FDA issued a warning letter to DynaPro International, Inc.
Recalls & Warnings
December 01, 2018
Seller of 5-HTP, Potassium & More Warned for Manufacturing Violations
On September 7, 2018, the FDA issued a warning letter to The Delano Company, Inc.
Recalls & Warnings
June 07, 2013
Maker of Colloidal Silver and Mushroom Extract Supplements Warned For Manufacturing Violations and Drug Claims
On May 2, 2013, the FDA issued a warning letter to Earthborn Products, Inc.
Recalls & Warnings
May 29, 2013
Seller of Colloidal Silver, Noni Juice and Noni Supplements Warned For Drug Claims
On May 16, 2013, the FDA issued a warning letter to Matrix Health Products, Inc.
Recalls & Warnings
February 14, 2013
Maker of Antioxidant and Anti-Aging Supplements Warned For Manufacturing Violations
On January 10, 2013, the FDA issued a warning letter to dietary supplement manufacturer Consolidated Marketing Unlimited, Inc.
Recalls & Warnings
May 17, 2013
Maker of Cardio, Arthritis, Cleanse Supplements and More Warned For Manufacturing Violations and Drug Claims
On May 8, 2013, the FDA issued a warning letter to Entrenet Nutritionals, Inc.
Recalls & Warnings
May 17, 2013
Hoodia, Sexual Enhancement Supplements and More Found To Be Adulterated, Misbranded
On March 01, 2013, the FDA issued a warning letter to Desert Rose Manufacturing, Inc.
Recalls & Warnings
February 24, 2011
Recall of Counterfeit Extenze Tablets Containing Drugs
On February 23, 2011, the FDA announced that Biotab Nutraceuticals, Inc. was notified that two lots of counterfeit product purporting to be Extenze contain undeclared drug ingredients. Specifically, lot 0709241 contains tadalafil and sildenafil, and lot 0509075 contains tadalafil and sibutramine.
Recalls & Warnings
August 28, 2008
FDA Finds Fault with Generic Toprol XL -- Problems Reported Earlier by ConsumerLab.com
On August 12, 2008, the U.S. FDA sent a letter to Sandoz Inc. warning of violations in its manufacture of Metoprolol Succinate ER tablets and other drug products. Sandoz's Metoprolol Succinate ER tablets are generic versions of Toprol XL.
Recalls & Warnings
March 28, 2011
FDA Cracks Down on Violators of Supplement Manufacturing Rules
In February and March 2011, the U.S. FDA posted Warning Letters sent, respectively, to a distributor of dietary supplements and a supplement manufacturer for violations of current Good Manufacturing (GMP) rules.
Recalls & Warnings
July 19, 2011
FDA Warns of Unsafe Drug in Slimming Supplements
On July 8, 2011, the U.S. FDA advised consumers not to purchase or use Slim Forte Slimming Capsule and Slim Forte Double Power Slimming Capsules. FDA laboratory analysis confirmed that these products contain sibutramine. Sibutramine is a controlled substance that was removed from the U.S.
Recalls & Warnings
September 13, 2010
ExtenZe Enhancement Supplements Seized in Canada
On August 19, 2010, Health Canada (Canada's health ministry) seized the sexual enhancement supplements "Male Enhancement ExtenZe" and "Women ExtenZe" which were imported from the U.S. Although legal and widely sold in the U.S.
Recalls & Warnings
September 16, 2015
Herbal Extracts Recalled
On September 15, 2015, Iowa Select Herbs, LLC issued a recall of numerous herbal exacts following a permanent injunction which required the company to stop selling supplements.
Recalls & Warnings
December 10, 2002
Company Ordered to Cease Deceptive Marketing of Weight Loss Product
A U.S. District Court in Texas has ordered Mark Nutritionals, maker of Body Solutions Evening Weight Loss Formula, to stop making certain claims with regard to the product. The order is in response to a complaint by the Federal Trade Commission (FTC) accusing the company of false advertising.
Recalls & Warnings
April 03, 2012
“Japan Rapid Weight Loss Diet Pills Green” Contains Hidden Drug Ingredient
April 3, 2012: The Food and Drug Administration (FDA) is advising consumers not to purchase or use "Japan Rapid Weight Loss Diet Pills Green," a product for weight loss sold on various websites, such as www.amazon.com, and distributed by Xiushentang.
Recalls & Warnings
September 11, 2015
Thirteen Manufacturers Ordered to Stop Selling Devil's Claw Supplements
On September 9, 2015, the New York State Attorney General ordered 13 manufacturers, including Thorne Research Inc., NBTY, Inc. (Puritan's Pride) Inc., and Now Foods, to stop selling devil's claw supplements which were found to contain the incorrect species of the plant.
Recalls & Warnings
September 23, 2014
Maker of Joint and Weight Products Warned for Hidden Drugs, Manufacturing Violations
On September 15, 2014, the FDA issued a warning letter to West Coast Laboratories, Inc., because the company's joint health supplements, Super ArthGold and Pro ArthMax, were found to contain hidden drugs.
Recalls & Warnings
April 09, 2003
Recall of Dangerous Sexual Enhancement Supplement Illegally Containing Viagra Ingredient
On April 4, 2003, the U.S. Food and Drug Administration's MedWatch program announced that Ultra Health Laboratories, Inc. and Bionate International, Inc. are warning consumers not to purchase or consume a product known as Vinarol tablets.
Recalls & Warnings
April 25, 2017
Beware of Products Which Promise to Treat or Cure Cancer, FDA Warns
On April 25, 2017, the FDA warned consumers to be aware of supplements and other products claiming to cure cancer.
Recalls & Warnings
July 01, 2003
Direct Marketers of Weight Loss, Impotence, and Arthritis Supplements Charged with Deceptive Claims
On July 1, 2003, the Federal Trade Commission (FTC) announced three enforcement actions against direct marketers of weight-loss products containing ephedra. The two settlements and one complaint, filed in U.S.
Recalls & Warnings
October 15, 2012
Recall of Nutrition Bars Containing Peanut Butter Due To Salmonella Risk
On October 11 and 12, 2012, four brands of nutrition bars were voluntarily recalled due to possible salmonella contamination of the peanut butter used in each of these bars. The peanut butter was produced by Sunland Inc., and is part of a larger recall of Sunland's peanut butter and peanut products.
Recalls & Warnings
October 24, 2012
Manufacturer of Green Tea, Vitamin E, Omega-3 and Cranberry Supplements Warned For Manufacturing Violations, Misbranding and Drug Claims
On October 16, 2012, the FDA issued a warning letter to dietary supplement manufacturer Advanced Nutritional Technology Inc.
Recalls & Warnings
May 03, 2013
Maker of Omega-3, Saw Palmetto, St. John’s Wort Supplements And More Warned For Manufacturing Violations and Drug Claims
On March 19, 2013, the FDA issued a warning letter to Sunset Natural Products Inc.
Recalls & Warnings
February 20, 2013
Cold, Flu and Stress Supplement Company Warned For Adulteration, Drug Claims And More
On October 5, 2012, the FDA issued a warning letter to dietary supplement manufacturer Wholistic Herbs, Inc. following a facility inspection which found the company's products, including At-Ease, Morning Calm, Aller-Ban, Stomach Flu, Kold & Koff, Kold Sore, and L.
Recalls & Warnings
March 01, 2013
Manufacturer of Joint, Weight Loss, Muscle Supplements and More Warned for Adulteration, Misbranding and Drug Claims
On February 4, 2013, the FDA issued a warning letter to DC Nutrition, Inc.
Recalls & Warnings
February 29, 2008
FTC Sues Sellers of Weight-Loss Pills for False Advertising
On February 8, 2008 the The Federal Trade Commission (FTC) charged a business operation with violating federal law by falsely claiming that its weight-loss pills cause users to lose weight without dieting or exercise.
Recalls & Warnings
September 08, 2018
Homeopathic Oral Sprays Recalled Due to Possible Bacterial Contamination
On September 5, 2018, Beaumont Bio Med, Inc., issued a recall of all of its water and alcohol-based homeopathic products because they have the potential to contain microbial contaminants.
Recalls & Warnings
January 18, 2002
Recall of Pepsin-containing Digestive Supplements
The U.S. Food and Drug Administration (FDA) released the following Class I recall information in its January 16, 2002 Enforcement Report.
Recalls & Warnings
March 14, 2011
22 Brands of Whey Protein Recalled Due to Salmonella Concern
The FDA posted a recall notice involving 22 brands of whey protein powder due to potential contamination with salmonella. The recall was voluntarily initiated on March 10, 2011 by the manufacturer of the products, Vitalabs, Inc.
Recalls & Warnings
July 05, 2012
FDA Warns Good Herbs Inc. of Manufacturing Violations and Drug Claims
The FDA issued a warning letter to Good Herbs Inc. on May 24, 2012, after a facility inspection found the company to be in violation of current manufacturing regulations, including lack of documentation and written procedures.
Recalls & Warnings
January 10, 2013
FDA Warns Aloe and Herbal Supplement Companies For Manufacturing Violations
AloeScience Labs, Inc. -- On November 14, 2012, the FDA issued a warning letter to AloeScience Labs, Inc.
Recalls & Warnings
April 19, 2007
Recall of Another Sex Enhancement Supplement
On April 17, 2007 the Food and Drug Administration (FDA) website posted an announcement that Jen-On Herbal Science International, Inc. is conducting a voluntary nationwide recall of the company's supplement product sold under the name H S Joy of Love. Jen-On Herbal Science International, Inc.
Recalls & Warnings
December 17, 2004
FDA Seizes Ginseng Because of Potentially Risky Pesticide Residues
On December 16, 2004, the Food and Drug Administration (FDA) announced that the U.S. District Court for the District of New Jersey issued a warrant for the seizure of imported ginseng held for sale at FCC Products, Inc., located in Livingston, N.J. The U.S.
Recalls & Warnings
October 12, 2005
Supplement Company to Pay $2 Million Penalty For Alleged Violations of FTC Order
On October 12, 2005 the Federal Trade Commission (FTC) announced that under the terms of a consent decree approved by the it for submission by the U.S. Department of Justice (DOJ) to the federal court for approval, NBTY, Inc. (NBTY, formerly Nature’s Bounty, Inc.
Recalls & Warnings
February 01, 2018
Seller of Supplements for Opiate Withdrawal Warned for Drug Claims
On January 11, 2018, the FDA sent warning letters to ten sellers of supplements promoted to treat opiate withdrawal following reviews of the companies' websites and social media which found statements and testimonials made about the products to be drug claims.
Recalls & Warnings
August 11, 2015
Company Ordered to Stop Making and Selling Supplements
On August 4, 2015, a federal court ordered a permanent injunction against dietary supplement company Atrium Inc., and two companies under the same ownerhship, Aspen Group Inc., Nutri-Pak of Wisconsin Inc.
Recalls & Warnings
October 06, 2011
Testosterone Booster Supplement Recalled for Containing Synthetic Steroid
On October 5, 2011, the U.S. FDA announced that Superior Metabolic Technologies Inc of Marietta, Georgia is voluntarily recalling all lots of the testosterone booster Uprizing 2.
Recalls & Warnings
January 02, 2012
FDA Warns XYMOGEN of Manufacturing and Labeling Violations Regarding Multiple Products
On December 13, 2011, the U.S. FDA sent a Warning Letter to Atlantic Pro Nutrients, Inc. (dba XYMOGEN) regarding labeling and/or manufacturing violations relating to its products, which include Borage CP-240, CoQmax CF, Immune Rx, Iron Glycinate, and Green Tea 600.
Recalls & Warnings
May 06, 2014
Two Weight Loss Supplements Found To Contain Drug
On May 5, 2014, the FDA warned consumers not to buy or use the weight loss supplements listed below because they were found to contain sibutramine.
Recalls & Warnings
October 10, 2013
Six Weight Loss Supplements Found to Contain Drugs
On October 10, 2013, the FDA advised consumers not to purchase or use the six weight loss supplements listed below because they were found to contain one or both of the undeclared drugs sibutramine and phenolphthalein.
Recalls & Warnings
February 06, 2015
Seller of Aloe Supplement Warned for Drug Claims
On January 21, 2015, the FDA issued a warning letter to Pristine Nutraceuticals, LLC, following a review of the company's website, http://www.digestaqure.com, which found statements made about DigestaCure to be drug claims.
Recalls & Warnings
November 30, 2013
"Diuretic" Weight Loss Supplement Recalled
On November 29, 2013, IQ Formulations issued a recall of all lots of diuretic weight loss supplement HYDRAVAX because one lot of the supplement was found to contain an undeclared prescription diuretic drug.
Recalls & Warnings
April 01, 2017
Maker of Prelief, Urinozinc Prostate Health Formula Warned for Manufacturing Violations
On March 16, 2017 the FDA issued a warning letter to DSE Healthcare Solutions, LLC, following a facility inspection which found the company's products, including Prelief and Urinozinc Prostate Health Formula to be adulterated because they were prepared, packed, or held under ...
Recalls & Warnings
February 02, 2018
Seller of Vitamin D, Omega-3's, Whey Protein & More Warned for Drug Claims
On January 24, 2018 the FDA issued a warning letter to Young Health Products, LLC following a review of the company's website which found promotional statements and testimonials made about the certain products, including Lugol's Solution 2%, Liquid Vitamin D3, Probimune, Flax Seed & Omega ...
Recalls & Warnings
October 18, 2005
FTC Stops False Claims about HGH Oral Spray
On October 18, 2005, the Federal Trade Commission (FTC) announced that, at its request, a federal court issued a temporary restraining order against marketers of oral sprays that supposedly contain human growth hormone (HGH) to stop them from making alleged false and deceptive claims and from ...
Recalls & Warnings
April 11, 2008
Twelve Dietary Herbal Supplements Recalled -- Possible Health Risk Associated with Ephedra, Aristolochic Acid and Human Placenta
On April 10, 2008, the FDA posted a recall notice issued the same day from Herbal Science International, Inc. (AKA Jen-On Herbal Science International, Inc.).
Recalls & Warnings
March 19, 2004
Seasilver Stopped from Making Claim to Cure "Over 650" Diseases
On March 17, the Food and Drug Administration (FDA) announced that Seasilver USA, Inc., and Americaloe, Inc.
Recalls & Warnings
May 20, 2014
FTC Charges Seller of Green Coffee Bean with False Weight Loss Claims, Fake Websites
On May 15, 2014, the Federal Trade Commission (FTC) filed a lawsuit against the sellers of Pure Green Coffee for making unsubstantiated weight loss claims and deceiving consumers with fake "news" websites.
Recalls & Warnings
April 17, 2015
Herbal Laxative and "Detox" Kit Containing High Levels of Lead and/or Arsenic Recalled in Canada
On April 15, 2015, Health Canada (the Canadian equivalent of the U.S. FDA) announced that St. Francis Herb Farm Inc.
Recalls & Warnings
October 17, 2013
Turmeric Spice Powder Recalled Due To High Lead Levels
On October 3, 2013, the OnTime Distribution Inc. issued a voluntary recall of PRAN brand spice powder Turmeric because it was found to contain high levels of lead.
Recalls & Warnings
December 27, 2012
Two Supplement Companies Warned For Manufacturing Violations
Sterling USA Neutraceutical Lab, LLC -- On November 16, 2012, the FDA issued a warning letter to Sterling USA Neutraceutical Lab, LLC following a facility inspection which found the company’s dietary supplements to be adulterated because they were have been prepared, packed, or held under ...
Recalls & Warnings
November 08, 2012
Chinese Herb Recalled Due To Undeclared Sulfites
On November 7, 2012, the New York State Department of Agriculture announced that Mayflower International Inc., aka Fu Xiang Yuan Trading Inc., is recalling Lily Dry after sample analysis revealed the product contains high levels of sulfites which were not declared on the label.
Recalls & Warnings
August 24, 2012
Protein Shots, Drinks and Gelatin Recalled For Potential Botulism Contamination
On August 17, 2012, Protica Inc.
Recalls & Warnings
June 01, 2012
Homeopathic Manufacturer Settles False Advertising Suit
June 1, 2012 — A federal court in California recently authorized the settlement of a class action lawsuit against Boiron Inc. and Boiron USA, Inc.
Recalls & Warnings
October 05, 2011
FDA Warns Nutrition Center Inc (Nutri-West) of Manufacturing Violations
The FDA published a Warning Letter dated September 9, 2011 to Nutrition Center Inc. (dba Nutri-West) informing the firm of manufacturing violations found during an inspection of the firm's facility in Douglas, Wyoming in April, 2011.
Recalls & Warnings
September 08, 2011
FDA Warns Several Supplement Manufacturers Not Following Good Manufacturing Practices
In recent weeks, the U.S. FDA has sent warning letters to several supplement manufacturers regarding deficiencies identified during audits of their facilities and not adequately corrected. Use the links below to access the Warning Letters on the FDA website.
Recalls & Warnings
April 21, 2011
FTC Targets Fake News Sites Making Deceptive Acai Claims
On April 19, 2011, the Federal Trade Commission (FTC) requested federal courts to temporarily halt the allegedly deceptive tactics of 10 operations using fake news websites to market acai berry weight-loss products.
Recalls & Warnings
September 13, 2010
Supplement for "Increasing Desire" Recalled for Containing Drug
The U.S. FDA posted a notice regarding the recall of Masxtreme Capsules by the distributor, Natural Wellness, Inc. FDA analysis has determined the product contains undeclared amounts of Aminotadalafil, an analog of tadalafil.
Recalls & Warnings
July 20, 2010
Recall of ED Supplement Containing Drug
On July 20, 2010, the U.S. FDA posted a news announcement from Good Health, Inc. regarding the recall of Vialipro, a sexual enhancement supplement sold nationally.
Recalls & Warnings
January 19, 2002
Recall of Certain Iron Supplements
The U.S. Food and Drug Administration (FDA) released the following Class III recall information in its January 16, 2002 Enforcement Report. A Class III recall is a situation in which use of or exposure to a violative product is not likely to cause adverse health consequences:
Recalls & Warnings
December 05, 2015
FDA Warns Companies Selling Products Containing Picamilon
On November 30, 2015, the FDA issued warning letters to five supplement companies selling products containing picamilon, which is not a lawful dietary supplement ingredient in the U.S.
Recalls & Warnings
April 26, 2015
FDA Identifies More Products Listing Synthetic Amphetamine
On April 22, 2015, the FDA issued warning letters to sellers of weight loss and workout supplements that list a synthetic, amphetamine-like compound called beta- methylphenylethylamine (BMPEA) on product labels.




