Product Reviews and Information for Chronic Obstructive Pulmonary Disease (COPD)
Search term may appear only in full report available to members. Join now for full access.
Clinical Update
12/03/2014
Vitamin D for COPD
A study of people with chronic obstructive pulmonary disease (COPD) found that taking vitamin D significantly reduced the risk of moderate to severe exacerbation, but only among people who began the study deficient in vitamin D. Get the details and information about other uses of vitamin D, along with our quality ratings of vitamin D supplements, in the Vitamin D Supplements Review >>
CL Answer
Which is the best pulse oximeter for home use?
Best pulse oximeter for home use (COVID-19, asthma and COPD). Differences between home use and FDA-approved medical pulse oximeters.

Product Review
Quercetin & Rutin Supplements Review
Quality's a Concern With Quercetin and Rutin Supplements -- Only 17% of Claimed Amount In One

Product Review
Muscle & Workout Supplements Review (Creatine and Branched-chain Amino Acids)
Do Creatine and BCAAs Really Improve Strength and Recovery?

CL Answer
Which is the best mask to prevent COVID-19 and how do cloth, disposable, N95, and KN95 masks compare? How can I stop glasses from fogging?
See our Top Picks for masks. Learn how to make COVID-19 masks from materials at home that can be almost as effective as surgical mask and N-95 masks.
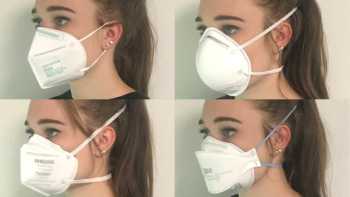
CL Answer
Are people with kidney disease at risk of getting too much potassium if they take a multivitamin? Which multivitamins are best for people with kidney disease?
Learn if people with kidney disease are at increased risk of getting too much potassium from multivitamins, find out which multivitamins do not contain potassium, and learn what other products should be avoided by people restricting potassium intake.

CL Answer
Dimethyl sulfoxide (DMSO): Health Effects & Safety Concerns
Dimethyl sulfoxide (DMSO) is a colorless liquid promoted for osteoarthritis, interstitial cystitis, tennis elbow, rotator cuff tendonitis, wound healing, and many other conditions. Find out if it works and if it's safe.

CL Answer
Can vitamins or other supplements cause a change in the ability to taste, or even a loss of taste?
Find out if supplements or vitamin deficiencies can cause changes in taste, loss of taste or smell. Information about vitamin D, zinc, vitamins A, B6, B12 and others. Plus medications and medical conditions that can alter taste and smell. ConsumerLab.com's answer explains.

Clinical Update
3/03/2021
Vitamin D for COPD and Asthma?
Does high-dose vitamin D help treat COPD or asthma? See what a new study found in the What It Does section of our Vitamin D Supplements Review. Also, see our Top Picks for vitamin D.
Clinical Update
11/11/2021
Multivitamins and Kidney Disease
People with chronic kidney disease are often advised to limit their intake of an ingredient that, they may not realize, is common in multivitamins. Learn more in the ConsumerTips section of the Multivitamin and Multimineral Supplements Review.
Clinical Update
5/31/2024
Have Kidney Disease? Watch What’s In Your Water
People with chronic kidney disease are particularly susceptible to the toxic effects of heavy metals. Even low levels of lead in drinking water may be harmful, according to a recent study. See our Water Filter Pitchers Review for details. Also see our Top Picks among water filter pitchers.
Clinical Update
3/15/2017
Anti-Inflammatory Effect of Juice Plus+?
Chronic systemic inflammation is a risk factor for cardiovascular disease, diabetes, and cancer. A recent clinical trial assessed the product Juice Plus+ for its ability to reduce this inflammation in obese men and women. The abstract of the published study indicates that it works, but the results tell a different story. For details, see the "What It Does" section of the Greens and Whole Foods Powders, Pills, and Drinks Review (which includes our tests of products) >>
Clinical Update
9/18/2020
Vitamin D for Failing Hearts?
Does high-dose vitamin D help or hurt people with chronic heart failure? Find out what a new study showed in the Cardiovascular Disease section of Vitamin D Supplements Review, which also discusses cholesterol, blood pressure and heart attack risk. Also see our Top Picks for vitamin D.
Clinical Update
7/26/2022
Vitamin D and Heart Failure
Does high-dose vitamin D improve heart function in people with chronic heart failure? Find out what a recent study showed in the Cardiovascular disease, blood pressure, and cholesterol section of our Vitamin D Supplements Review.
Clinical Update
12/09/2022
Allulose Questions
- Is allulose (a low-calorie sweetener) likely to form compounds that have been linked with chronic disease?
- How can I avoid allulose made from genetically modified corn?
Clinical Update
2/10/2023
Magnesium and Artery Calcification
Did magnesium supplementation slow coronary artery calcification in people with chronic kidney disease? See what a recent study found in the What It Does section of our Magnesium Supplements Review, which includes our Top Picks for magnesium.
Also see: Which supplements or foods can help lower cholesterol and keep my heart healthy? Are there any to avoid?
Clinical Update
5/19/2023
Caution With Sweeteners
Certain sweeteners should not be used for decreasing weight or preventing chronic diseases, according to a new report. Find out which sweeteners are of concern and why in our updated article about sweeteners.
Clinical Update
2/21/2024
Lumbrokinase for Cancer, Infection?
Can lumbrokinase help slow cancer progression or treat chronic infections such as Lyme disease? Find out in our article about lumbrokinase.
Clinical Update
4/24/2025
Microplastics Linked with High BP, Stroke
Exposure to higher amounts of microplastic was associated with increased risk of several chronic diseases, according to a recent study. Get the details in article about Microplastics in water, salt, foods, and beverages. Also see our Water Filter Pitchers Review for our Top Pick for removing microplastics.
News Release
April 15, 2026
Think Expensive NAC Is Better? ConsumerLab Tests Say Otherwise
White Plains, NY, April 15, 2026 — NAC (N-acetyl cysteine) supplements are widely marketed for “detoxification,” immune support, and respiratory health, as NAC is converted in the body into the antioxidant glutathione.
Recalls & Warnings
June 20, 2003
FTC Charges Marketers of Seasilver with False and Deceptive Claims
On June 19, 2003, The Federal Trade Commission (FTC) and the Food and Drug Administration (FDA) announced coordinated actions against two companies - both charged with promoting the dietary supplement "Seasilver" with unsubstantiated medical claims. The agencies' actions against Seasilver USA, Inc.
Recalls & Warnings
May 08, 2021
Seller of Immune Bio Green Cell Warned by FDA
On March 30, 2021, the FDA issued a warning letter to Immune & Genetics Protocols, LLC following a review of the company's websites, which found statements made about the company's product Immune Bio Green Cell to be drug claims.
Recalls & Warnings
April 18, 2022
Moringa Concern: Blood Clots
A 63-year-old woman in New York with type 2 diabetes developed a blood clot in the lungs (i.e. pulmonary embolism) that her physicians suspect may have been caused by the use of a supplement containing Moringa oleifera extract (Ebhohon, Int J Emerg Med 2022).
Recalls & Warnings
March 31, 2005
FDA Warns Marketer of "Vitamin O" Product to Cease Unsubstantiated Claims
The U.S. Food and Drug Adminstration (FDA) has sent a Warning Letter (dated February 8, 2005) to Donald L. Smyth, President, R-Garden Inc., and Rose Creek Health Products, Inc. warning that its "Vitamin O" product was, among other things, being marketed with unsubstantiated health claims.
Recalls & Warnings
December 17, 2020
FTC Crackdown on Six Deceptive CBD Products
On December 17, 2020, the FTC announced that it is taking action against six companies for selling CBD products with unsupported and deceptive health claims that they can treat a variety of conditions, including cancer, heart disease, hypertension, Alzheimer's disease, and others.
Recalls & Warnings
August 30, 2012
Supplement Company Warned For Medical Claims and Misbranding Of Omega-3, CoQ10, Noni Juice And More
On July 12, 2012, the FDA issued a warning letter to Alfa Vitamins Laboratories, Inc.
Recalls & Warnings
April 11, 2013
Glutathione Supplement Maker Warned For Manufacturing Violations and Drug Claims
On April 5, 2013, the FDA issued a warning letter to The Glutathione Corporation following a facility inspection which found the company's supplements, including Ultrathione 500 Glutathione, Ultrathione 1000 Sports Pack, Ultrathione Performance, Ultrathione Extreme Performance, Ultrathione Health ...
Recalls & Warnings
April 17, 2020
Seller of Vitamin D, MSM, Selenium & More Warned for Drug Claims
On March 31, 2020, the FDA issued a warning letter to KetoKerri LLC following a review of the company's website, which found statements made about some of the company's products, including Dr.
Recalls & Warnings
July 29, 2009
FDA Warns Against Body Building Supplements with Steroid-like Compounds
On July 28, 2009, the U.S. FDA notified the public about new safety information concerning products marketed for body building and increasing muscle mass.
Recalls & Warnings
April 02, 2019
FDA Issues Strong Warnings to Sellers of CBD
On March 28, 2019, the FDA issued warning letters to three companies for making unsubstantiated claims about the health benefits of CBD (cannabidiol) products on their websites.
Recalls & Warnings
September 13, 2013
Metagenics Warned For Misbranding of Medical Foods and Drug Claims
On August 13, 2013, the FDA issued a warning letter to Metagenics, Inc.
Recalls & Warnings
June 11, 2015
FDA Warns Maker of Fruit Energy Drinks for Drug Claims
On May 19, 2015, the FDA issued a warning letter to CK Management, Inc. following a facility inspection which found statements made on product labels and websites about Whole5, Fruit of the Spirit and ViaViente "whole food" fruit puree energy drinks to be drug claims.
Recalls & Warnings
August 08, 2017
Study Shows Severe Risk in Drinking Hydrogen Peroxide
An analysis of records from the National Poison Data System from a 10-year period ending in 2011 identified 294 reported incidents of people developing adverse symptoms after ingesting 35% (high-concentration) hydrogen peroxide (H202). Among these, 41 (13.
Recalls & Warnings
July 12, 2017
More Bodybuilding Supplements Recalled
On July 11, 2017, Andropharm recalled two bodybuilding supplements, Sten Z and M1 Alpha, because these products contain derivatives of anabolic steroids.
Recalls & Warnings
June 20, 2017
Beware of Bodybuilding Supplements, FDA Warns
On June 20, 2017, the FDA warned consumers to beware of bodybuilding products because a number of these products have been found to contain steroids or steroid-like substances. Such products could cause serious and even life-threatening health risks, including:
Recalls & Warnings
March 16, 2006
Garden of Life, Maker of Primal Defense, Settles FTC Charges
On March 9, 2006, The Federal Trade Commission (FTC) announced that an operation that marketed dietary supplements sold at Whole Foods Market, GNC, the Vitamin Shoppe, and on the Internet settled FTC charges that they made deceptive advertising claims about their supplements.




