Our Members Asked:
I read that famotidine (Pepcid) may help treat COVID-19. Is this true? Might a proton pump inhibitor, like Prilosec, help?
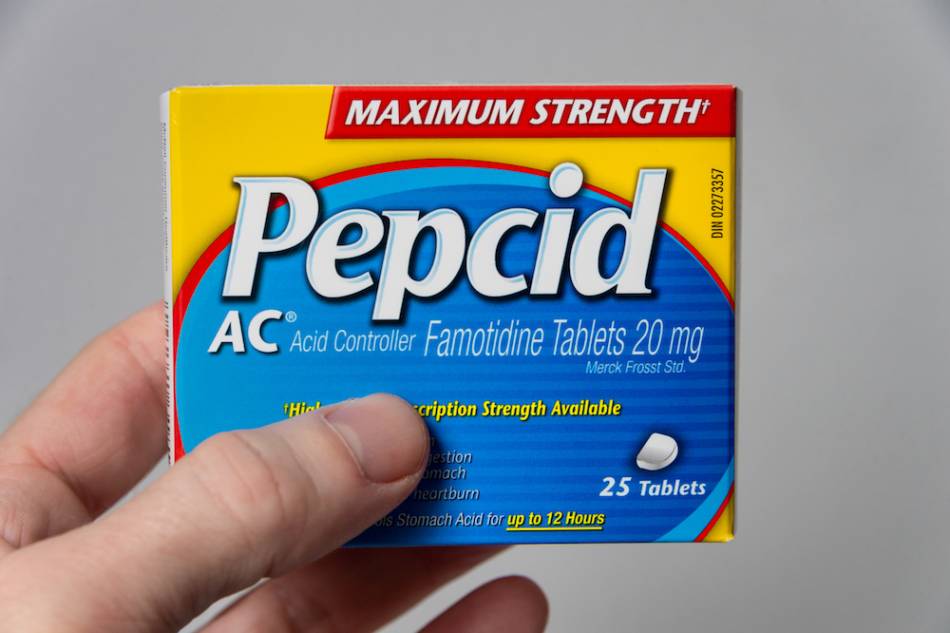
Answer:
Interest in famotidine for treating COVID-19 (caused by the coronavirus SARS-CoV-2 infection) was triggered by a preliminary report that people in China with COVID-19 who had taken famotidine for its approved use (as an "H2 blocker" to reduce stomach acidity) had better outcomes than people who were not taking it or who had been using other types of acid reducers, such as Prilosec (omeprazole — a proton pump inhibitor or PPI).
However, famotidine has not been shown to prevent SARS-Cov-2 infection and although some preliminary studies and reports suggested a possible benefit of famotidine in people with COVID-19, a large study of hospitalized COVID-19 patients given famotidine upon admission found no benefit.
PPI use may increase the risk of some microbial infections, but it does not seem to affect the risk of SARS-Cov-2 infection. Preliminary evidence has found PPI use to be associated with increased risk of COVID-19 being severe, although not with death.
The evidence regarding famotidine and PPIs in COVID-19 treatment:
An article in Science (April 26, 2020) that discusses the findings from China stated: "In reviewing 6212 COVID-19 patient records, the doctors noticed that many survivors had been suffering from chronic heartburn and were on famotidine rather than more-expensive omeprazole (Prilosec), which is a proton pump inhibitor and the medicine of choice both in the United States and among wealthier Chinese. Hospitalized COVID-19 patients on famotidine appeared to be dying at a rate of about 14% compared with 27% for those not on the drug, although the analysis was crude and the result was not statistically significant." The article also notes anecdotal reports in the U.S. of a women who recovered from symptoms of COVID-19 the day after taking a "megadose" of oral famotidine and of "dramatic improvements" in five other sick people (three confirmed for COVID-19) after taking oral famotidine.
A review of the records of COVID-19 patients (none of whom required immediate mechanical ventilation) admitted to Columbia University hospital in New York showed that those who had received famotidine (intravenous or oral, to suppress gastric acid) within 24 hours of admission were 70% less likely to have died within 30 days of admission than those who had not received famotidine. There was no association between famotidine and the need for intubation (being put on a ventilator). Among the patients who had received famotidine, 72% had received it orally (10 to 40 mg daily) and 28% intravenously (dose not reported) (Freedberg, Gastroenterology 2020).
However, a larger study of more than 400 hospitalized COVID-19 patients given famotidine (averaging about 27 mg daily) upon admission found no benefit compared to patients not given famotidine. In fact, those not receiving famotidine at home but receiving it in the hospital had a 77% higher risk of death within 30 days (Yeramaneni, Gastroenterol 2021).
Interestingly, in the Columbia study, use of PPIs, which more potently suppress gastric acid, was not associated with reduced risk for death or intubation. Subsequent analysis of data from nine observational studies (including the Columbia study) actually found that PPI use was associated with a 67% increased risk of severe disease in people with COVID-19, as well as an increased risk of secondary infections, although it was not associated with an increased risk of death (Yan, MedRxiv — preprint).
A case study that followed ten adults (ages ranging from 20s to 70s) with COVID-19 who gave themselves high-dose famotidine (ranging from 20 mg to 80 mg three times daily, with most taking the highest dose) for about 5 to 21 days (median of 11 days) found that all reported marked improvements in COVID-19-related symptoms within 24 to 48 hours of starting the drug, and none required hospitalization. Cough and shortness of breath improved more rapidly than fatigue. The time period between onset of symptoms and starting famotidine ranged from 2 days to 26 days. Famotidine was well tolerated: 7 patients did not experience any adverse events, two reported dizziness, and one reported gastrointestinal side effects. These are known side effects of famotidine, as discussed below. One patient experienced temporary forgetfulness. All side effects resolved on discontinuation of famotidine (Janowitz, Gut 2020).
Based on computational modeling, it was proposed that famotidine might inhibit an enzyme (3-chymotrypsin-like protease or 3CLpro) essential for processing proteins needed for replication of the SARS-CoV-2 virus (Wu, Acta Pharm Sin B 2020). However, subsequent laboratory research showed that famotidine does not inhibit this enzyme or viral replication (Loffredo, bioRxiv — preprint).
A clinical trial in New York began in April of 2020 in which famotidine was given intravenously in very large doses to patients hospitalized with COVID-19. Patients received three intravenous doses daily, each providing 120 mg of famotidine, i.e., 360 mg total per day. (Note: A normal oral daily dose of famotidine for heartburn is 10 to 20 mg up to twice daily.) The study was completed in September, but clinical trial results have yet to be published.
Until results of this or other trials with famotidine are published, there is no solid evidence that famotidine is effective in treating COVID-19.
The evidence regarding famotidine and PPIs in COVID-19 prevention:
At this time, there is not even anecdotal evidence that famotidine prevents COVID-19. Although preliminary evidence suggested that PPIs (which also reduce stomach acid, although in a different way than H2 blockers like famotidine) might increase the risk of COVID-19 infection, subsequent data indicated that PPIs neither increase nor decrease the risk.
One study found once-daily use of PPIs — but not H2 blockers like famotidine — to be associated with a 115% increase in the risk of contracting COVID-19 in comparison to not taking PPIs. Furthermore, there was a 267% increase in risk when PPIs were taken twice daily. The results were based on a national phone survey of 53,130 Americans in May and June 2020. The researchers noted that PPI use is associated with increased risk for other enteric infections, likely because PPIs dramatically reduce stomach acidity that can deactivate bacteria and viruses. For example, a related coronavirus (SAR-CoV-1) is deactivated at a pH of less than 3.0, but twice daily PPI use increase the 24-hour median pH of the stomach to greater than 6. As twice daily use of PPIs has not been shown to be more effective than once-daily use, the researchers cautioned that PPIs should only be used when necessary and at the lowest effective dose (Almario, Am J Gastroent 2020).
However, analysis of data from seven observational studies, including the one above, found that proton pump inhibitor use was not associated with the risk of COVID-19 infection, regardless of where the study was conducted (i.e., western countries or Asian countries) (Yan, MedRxiv — preprint).
Be aware of some risks with famotidine:
Side effects that have been reported with famotidine at a dose of 40 mg at bedtime (taken to treat acute gastrointestinal ulcers) are headache (4.7%), dizziness (1.3%), constipation (1.2%), and diarrhea (1.7%) (Pepcid insert, Drugs@FDA). The study in New York excluded COVID-19 patients with decreased kidney function because high doses of famotidine can cause heart problems in them. Other adverse effects that have been infrequently reported with famotidine use include including jaundice and anorexia; rare cases of blood abnormalities; musculoskeletal pain; seizures; psychiatric disturbances; respiratory reactions (bronchospasm and interstitial pneumonia); various skin effects maladies including, very rarely, epidermal necrosis; tinnitus; and taste disorder. In addition, famotidine should not be used by anyone with an allergy to famotidine or other acid reducers.
Long-term use of famotidine and other medications that lower stomach acidity has been associated with deficiency in vitamin B12 and zinc, possibly due to poorer extraction of these nutrients from foods. Supplementation with these nutrients may be advisable with long-term use of famotidine. When discontinuing famotidine, a rebound in gastric acid production can potentially occur, causing abdominal discomfort. To help avoid this, do not stop abruptly, but taper the dose down over several days.
Also be aware that some over-the-counter formulations of famotidine include substantial amounts of other active ingredients. Pepcid Complete, for example, provides a modest amount of famotidine (10 mg) along with 320 mg of calcium and 70 mg of magnesium per tablet, and no more than two should be taken per day. Excessive intakes of calcium or magnesium from supplements are not advisable.
Join today to unlock all member benefits including full access to all CL Answers and over 1,400 reviews.
Join NowAlready a member? Sign In Here.
Join now at www.consumerlab.com/join/












Submit your comment
This feature is restricted to active members.
Join now to add comments and get all member benefits, including over 1,400 reviews.
Join NowAlready a member? Sign in here.