Reviews and Information for Green Tea
Green Tea Supplements and Drinks
If you want to find the best green tea high in antioxidants such as EGCG, you'll want to see our laboratory findings. We discovered a shocking range of EGCG, from 8 mg to more than 450 mg per serving! See results for popular brands of green tea as tea bags, loose leaf tea, bottled tea, ceremonial matcha powder, and green tea extract supplements. Also see how much caffeine we found, compare prices, and learn what conditions green tea is good for and about potential side effects.
Green Tea Supplements and Drinks ReviewSearch term may appear only in full report available to members. Join now for full access.
CL Answer
What are the health benefits of green tea, and is it safe?
ConsumerLab explains green tea's potential health benefits for everything from fighting cancer to helping your heart.

Product Review
Weight Loss Supplements Review (7-Keto DHEA, Forskolin and Stimulant Blend Supplements)
Choose the Best Weight Loss Supplement. Be Careful With Weight Loss Supplements — Few Pass Quality Testing and Safety Review.

Product Review
Fruits, Veggies, and Other Greens Supplements Review (Including Spirulina and Chlorella)
Avoid Lead in Greens, Problems with Pills, and Don't Give Up Eating Whole Foods.

News Release
February 26, 2025
ConsumerLab Survey Shows 21 Most Popular Vitamins and Supplements
White Plains, New York, February 26, 2025 — A recent survey of more than 8,400 people who regularly use dietary supplements shows that among the 212 most popular supplements, those that experienced the greatest absolute growth in popularity during 2024 were biotin (+9.
News Release
February 26, 2024
Latest ConsumerLab Survey Shows Growth in Popularity of Magnesium and Several Smaller Supplements
White Plains, New York, February 26, 2024 — A recent survey of more than 10,000 people who regularly use dietary supplements shows that supplements that experienced the greatest absolute growth in popularity during 2023 were pregnenolone (+7.5 percentage points), magnesium (+4.8 pts), berberine (+4.
Clinical Update
9/16/2024
Green Tea & Iron Deficiency
Severe iron-deficiency anemia was recently reported after regular consumption of green tea. Get the details in our Green Tea Review, which includes our Top Picks for green tea.
Also be aware that drinking green tea may significantly affect blood levels of a common beta-blocker, as well as statins, blood-thinners, and thyroid medication. See the Concerns and Cautions section of our Green Tea Review.
Clinical Update
6/03/2015
Green Tea for Weight Control?
Many studies have looked at whether green tea can help with weight management. The results have been mixed. A recently published study used a high-dose green tea extract. Although the researchers concluded it worked, a closer look suggests otherwise, and points to a potential risk. For details, as well as the results of our tests of green tea supplements, beverages, and brewable green tea, see the Green Tea Review >>
Clinical Update
4/13/2014
Does Green Tea Improve Memory?
This week, news sources reported that a study found green tea helps working memory. While green tea may have benefits, a closer look at this study suggests the news was exaggerated and misleading. For details about the study (and information about other effects of green tea and our tests of green tea supplements, teas, and drinks) see the update in the Green Tea Supplements, Drinks and Teas Review >>
Clinical Update
12/17/2019
Green Tea and Breast Cancer Risk
Is there a relationship between green tea consumption and breast cancer risk? Learn what a large recent study showed in the Cancer Prevention section of the Green Tea Review. Also see our Top Picks for green tea -- including brewable green tea (from bags and loose tea) and green tea drinks.
Product Review
Multivitamin and Multimineral Supplements Review
Best Multivitamins -- Caution with Gummies

Product Review
Prostate Supplements Review (Saw Palmetto and Beta-Sitosterol)
Find the Best Prostate Supplement for Symptoms of BPH. See Which Prostate Supplement Passed or Failed CL's Tests of Quality.
CL Answer
I take lisinopril (Zestril), an ACE inhibitor drug to lower blood pressure. Are there supplements I should avoid, or be taking, due to this drug?
When taking ACE inhibitors, find out which supplements should be avoided. Interaction info on Lisinopril or another ACE inhibitors.
Clinical Update
8/25/2015
Are Green Tea Pills Safe?
Studies have suggested that extracts of green tea can reduce the risk of certain cancers, improve memory, shrink uterine fibroids, and even help with weight reduction. However, liver toxicity has been a concern with green tea extracts. A large, government-funded study recently found that pills of green tea extract can be safe for many people, but some may experience adverse reactions, including elevated liver enzymes. Get the details in the "Concerns and Cautions" section of the Green Tea Review, along with information about what green tea does and our tests of green tea extracts, brewable teas, and beverages >>
CL Answer
Do any supplements naturally suppress appetite and help with weight loss?
Learn which natural ingredients can suppress appetite and find if these natural appetite suppressants help with weight loss.

Clinical Update
8/09/2015
Green Tea May Inhibit Starch Absorption
A study recently found that taking a green tea extract along with a starchy meal (corn flakes) significantly reduced carbohydrate absorption. This may help explain why green tea may help modestly with weight management. Details about this study, including dosage, along with our tests of green tea supplements and teas, are found in the Green Tea Review »
Clinical Update
1/18/2014
Green Tea "Blocks" Beta-Blocker
A new study shows that drinking green tea can drastically reduce the amount of a beta-blocker (nadolol, Corgard) absorbed into the bloodstream. Beta-blockers are typically taken to lower blood pressure, raising concern for people using green tea along with this beta-blocker and, possibly, related medications. Get the details, along with our tests of green tea drinks and supplements, in the update to the Green Tea Review >>
If you take a beta-blocker (which includes the popular drugs metoprolol (Lopressor, Toprol) and propranolol (Inderal)), be aware that chromium and CoQ10 may offer benefits.
Clinical Update
6/20/2014
Green Tea and Liver Toxicity
Green tea extract was listed as one of the most common dietary supplements linked to liver injury in a newly published guideline for diagnosing and treating drug-induced liver injury. Find out what one of the authors of the guideline had to say about the catechin content of some green tea supplements, and get our test results showing the amounts of catechins in green tea supplements in the Green Tea Supplements, Drinks and Brewable Teas Review >>
Clinical Update
4/02/2019
Green Tea and Breast Cancer
Drinking green tea has been associated with a reduced risk of breast cancer recurrence. But what effect does high-dose green tea extract have? See what a major study found in the What It Does section of the Green Tea Review. Also see our Top Picks among green tea products.
Clinical Update
6/30/2020
Getting the Most from Green Tea
Our Green Tea Review shows that matcha green tea provides higher amounts of healthful catechins than regular green tea. This week, we added information about why this is as well as the results of a new study showing which type of matcha provides the highest levels of polyphenols (i.e., catechins). For details see the What to Consider When Buying section of the review. Also see our Top Picks among green tea products.
Clinical Update
8/27/2020
Is Expired Green Tea Safe?
We were asked if it is safe to use green tea past its "best by" date and if its flavor or health benefits would be diminished. See our answer in the How to Use section of our Green Tea Review. Also see our Top Picks for green tea — including tea bags, loose tea, matcha, and green tea supplements.
Clinical Update
1/06/2025
Green Tea Interaction for Men
Green tea may significantly increase blood levels of a common medication for men. Get the details in the Concerns and Cautions section of our Green Tea Review, which includes information about many other green tea interactions, including with statins and blood-thinners.
Also see our Top Picks among green tea beverages and supplements.
Clinical Update
5/29/2025
"Forever Chemicals" In Green Tea?
Testing by a separate research group identified detectable levels of PFAS ("forever chemicals") in one-third of popular brands of green tea, according to a recent report. Get the details, and see our take on the evidence, in the Concerns and Cautions section of our Green Tea Supplements Review, which includes our Top Picks for green tea bags, matcha powder, and green tea supplements.
CL Answer
How significant are the risks to drinking tea from China from heavy metals?
Find out which types of tea from China pose the greatest risk of toxic heavy metal contamination, including green tea.

CL Answer
Does coffee have brain benefits?
People commonly drink coffee to increase alertness, counteract fatigue, and boost cognition, but does it really improve memory and cognitive function? Find out, and learn about risks linked with drinking too much coffee.

News Release
February 15, 2024
Unveiling the Truth About Green Tea’s EGCG Content
White Plains, NY, February 15, 2024 — ConsumerLab.com, a trusted name in health and wellness, sheds light on the EGCG (epigallocatechin gallate) content in green tea.
CL Answer
Do any supplements help with gum disease or periodontitis?
Supplements for healthy gums may help with gum disease or periodontitis. Learn more about omega fatty acids, lycopene, and probiotics.

Recalls & Warnings
October 09, 2025
Lipton Green Tea Citrus Recalled
On September 17, 2025, PepsiCo, Inc.
Clinical Update
3/10/2020
Staying Safe With Green Tea
If you use a green tea extract (alone or as part of a supplement formula), it is important to take it with food according to several reports, as explained in the Concerns and Cautions section of the Green Tea Review. Also see our Top Picks for green tea in bags, loose, as matcha powder, and in other forms.
Clinical Update
11/25/2017
How Hot Should Green Tea Be?
Drinking green tea may reduce the risk of certain cancers, but its effect on gastric (stomach) cancer may depend on the temperature at which you drink it, according to a new study. Find out what the best temperature seems to be in the "What It Does" section of the Green Tea Review >> (Also see how brands of green tea, including matcha, stack up in our tests.)
Clinical Update
11/12/2019
Green Tea & Stroke
Studies have found green tea consumption to be associated with a decreased risk of stroke. However, a recent study found this to be the case for only for one gender. For details, see the What It Does section of the Green Tea Review. Also see our Top Picks for green tea.
Clinical Update
8/17/2019
Green Tea and Blood Sugar
Can drinking green tea reduce the rise in blood sugar after a meal, and if so, is the effect greater in the morning or evening? See what a new study found in the What It Does section of the Green Tea Review. Also see our tests and Top Picks among green tea supplements, and brewable and bottled teas.
Clinical Update
9/24/2019
Does Green Tea Prevent Cognitive Decline?
Does drinking green tea reduce the risk of cognitive decline and, if so, how much do you have to drink? A multi-year study addressed this question. See the results in the Memory and Cognition section of the Green Tea Review. Also see our Top Picks for green tea.
Clinical Update
1/30/2019
Getting the Most from Green Tea
If you drink green tea to get beneficial compounds like EGCG, learn about a recent study that suggests that the type of water you use to brew tea drastically affects the amount of EGCG you get. Get the details in the How to Take section of the Green Tea Review. Also see our Top Picks for green tea.
Clinical Update
3/30/2019
Green Tea for Lowering Blood Pressure
Drinking green tea can help lower high blood pressure — and even reverse ventricular hypertrophy, according to a new study. For details, including the amount and brand of tea used, see the What It Does section of the Green Tea Review. Also see our Top Picks for green tea.
Clinical Update
9/12/2020
Green Tea and Cholesterol
Can green tea help lower cholesterol levels? Find out what a recent study showed in the What It Does section of Green Tea Review. Also see our Top Picks for green tea.
Clinical Update
9/29/2020
Green Tea for Memory?
Can a green tea extract supplement improve memory or cognitive function in healthy, older men and women? Find out what a recent study showed in the What It Does section of our Green Tea Review. Also see our Top Picks for green tea supplements and drinks.
Clinical Update
1/16/2015
Another Use for Green Tea?
A recently published study found that rinsing with green tea reduced plaque-forming bacteria in the mouth just as well as a prescription mouthwash. Get the details, including the form and dose of green tea used, in the Green Tea Supplements, Drinks and Brewable Teas Review >>
Clinical Update
4/22/2015
Green Tea Extract and Prostate Cancer
A new study, funded by the National Institutes of Health, showed that fewer men with pre-cancerous lesions developed prostate cancer if given a daily green tea extract supplement, although the results were not statistically significant due, in part, to the small size of the study. However, a somewhat similar study found a significant benefit. Learn more about these studies, and see our test results for green tea supplements, teas, and drinks, in the Green Tea Review >>
Clinical Update
6/18/2014
Does Green Tea Really Help With Weight?
A new study found that a popular green tea extract did not cause a reduction in weight compared to placebo, despite the fact that some previous studies have shown a benefit. Get more details, including which green tea extract was used, in the update to the Green Tea Supplements, Drinks and Teas Review >>
Clinical Update
9/28/2013
Green Tea to Treat Fibroids
A new study suggests that green tea extract may help shrink uterine fibroids (common in women of childbearing age) and reduce symptoms. For details, as well as test results and comparisons of green tea extracts and brewable teas and drinks, see the updated Green Tea Review >>
Clinical Update
12/23/2013
Liver Toxicity with Supplements, Including Green Tea
An increase in cases of liver toxicity associated with dietary supplement use has recently been reported. In an article about the findings, The New York Times reports that bodybuilding supplements and products containing green tea extracts are among those associated with such cases. For more information about green tea extracts and toxicity, see the Concerns and Cautions section of the Green Tea Supplements Review >>
Clinical Update
7/11/2015
Green Tea for Weight Management?
Green tea has been touted for weight management, but a recent study with a green tea extract casts doubt. More details are found in the "What It Does" section of the Green Tea Review >>
Clinical Update
7/06/2016
Lower Risk of Death With Green Tea
A long-term study of men in China found that those who regularly drank green tea — particularly larger amounts — were less likely to die during the study and had lower risks of dying from cardiovascular disease or cancer, compared to non-green tea drinkers. Get the details in the "What It Does" section of the Green Tea Review >>
Clinical Update
3/16/2016
Don't Take Green Tea With Iron?
A study shows that iron can bind to a key flavanol (EGCG) in green tea, causing it to lose its anti-oxidant activity. Should you avoid iron-containing foods when using green tea? Find out in the "What to Consider When Using" section of the Green Tea Supplements and Teas Review >>
Clinical Update
6/18/2016
Green Tea & Cognition
Green tea consumption has been associated with a lower prevalence of cognitive impairment in older adults. However, a recent study evaluating the effects of a green tea powder on older adults with Alzheimer's disease and other forms of dementia failed to show a benefit. For more information, see the "What It Does" section of the Green Tea Review >>
Clinical Update
3/07/2018
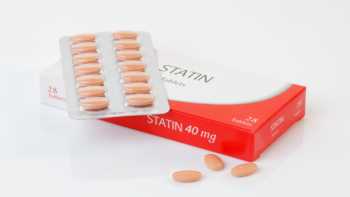
Statins and Green Tea
If you take a statin drug to lower your cholesterol, be aware that green tea can moderately affect blood levels of certain statins, as noted in a recent report. This effect also depends on the amount of green tea consumed and individual variability. For details see the Concerns and Cautions section of the Green Tea Review.
Clinical Update
3/27/2018
Green Tea and Ovarian Cancer
Drinking green tea was associated with a reduced risk of developing ovarian cancer in a recent analysis. Get the details about this and green tea's relationship to other types of cancer in the What It Does section of the Green Tea Review. (Also see our test results for various types of green tea, including matcha, bagged tea, loose tea, and supplements.)
Clinical Update
7/20/2018
Green Tea as a Prebiotic
Green tea may have a prebiotic effect — stimulating the growth of beneficial bacteria in the gut. Find out what clinical trials are showing in the What It Does section of the Green Tea Review. (Also see our Top Picks among green tea products).
Clinical Update
10/16/2018
A Caution With Green Tea
Although consumption of green tea is generally associated with a reduced risk of cancers, drinking it a certain way may increase the risk of certain cancers. Get the details in the Cancer Prevention section of the Green Tea Review. (Also see our Top Picks among green tea in bags, bottles, supplements, and matcha.)
Clinical Update
3/11/2022
Green Tea and Colon Polyps
Can taking green tea extract reduce the recurrence colon polyps? See what a recent study found in the What It Does section of our Green Tea Review. Also see our Top Picks among green tea extracts, tea bags, loose tea, and matcha powders.
See our answer to the question: Which supplements help reduce the risk of colorectal cancer?
Clinical Update
3/12/2024
Green Tea & Blood Sugar
Does green tea reduce fasting blood sugar levels? See what a recent study showed in the Blood Sugar Control/Diabetes section of our Green Tea Review, which includes our Top Picks for green tea.
Clinical Update
3/22/2024
Lower Cholesterol with Green Tea
Drinking green tea can lower cholesterol or triglyceride levels, as was recently shown. See the details in the What It Does section of our Green Tea Review, which includes our Top Picks for green tea.
Clinical Update
2/03/2023
Green Tea & Heart Disease
Find out how many cups of green tea per day is associated with lower risk of heart disease in the What It Does section of our Green Tea Supplements Review, and see our Top Picks for green tea.
Clinical Update
2/14/2023
Green Tea: More EGCG and Less Caffeine?
A CL member asked how steeping green tea in cold water rather than brewing in hot water affects levels of polyphenols and caffeine in the tea. See what a study found in the ConsumerTips section of our Green Tea Review, which includes our Top Picks for green tea.
Clinical Update
7/25/2023
Green Tea for Fibroids?
A preliminary clinical study showed that EGCG from green tea extract may help shrink uterine fibroids. New research suggests why. For details, see the Fibroids section of our Green Tea Supplements Review. Also see our Top Picks among green tea supplements and teas and find out how much EGCG they contain.
Clinical Update
9/01/2023
Green Tea Interactions
Drinking green tea can affect the absorption of many drugs, as was most recently shown with raloxifene (Evista), a medication used to treat osteoporosis and reduce breast cancer risk. Learn about these interactions (including those with certain statins, beta-blockers, and blood thinners) in the Concerns and Cautions section of our Green Tea Review. Also see our Top Picks among green tea products.
Clinical Update
9/12/2023
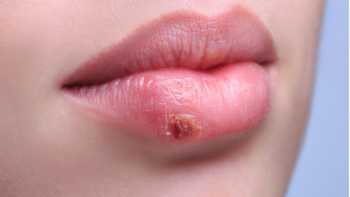
Green Tea for Cold Sores?
Does green tea help prevent or treat cold sores? Find out what research suggests in our Green Tea Review, which includes our Top Picks for green tea.
Also see: Can any vitamins or minerals help prevent or reduce canker sores or cold sores?
Clinical Update
8/22/2023
Green Tea and Blood Sugar
A laboratory study showed how green tea might help control blood sugar. Get the details in the Blood Sugar Control/Diabetes section of our Green Tea Review, which includes our Top Picks for green tea.
Also see: Which supplements and foods can help lower or control blood sugar?
Clinical Update
11/21/2023
Green Tea for Blood Cancer?
Does supplementing with green tea improve biomarkers in patients with leukemia, lymphomas or other blood cancers? Find out what research has shown in the Cancer Treatment section of our Green Tea Supplements Review, which includes our Top Picks for brewable tea and green tea supplements.
Product Review
Protein Powders, Shakes, and Meal Replacements Review
Find Out Which Protein Products Passed or Failed Our Tests

CL Answer
Is there a risk of liver toxicity with certain supplements?
Find out if there is risk of liver damage from supplements such as green tea, niacin, red yeast rice and vitamin A.
CL Answer
Which supplements and foods reduce or increase the risk of kidney stones?
Find out which supplements, foods and beverages may increase or decrease the risk of kidney stones, including information about potassium, calcium, choline, turmeric and curcumin, vitamin C, as well as foods and beverages such as green tea, lemon juice, orange juice, cranberry juice, chocolate and almonds.

CL Answer
Do any supplements, foods or lifestyle modifications help with brain function, like memory and cognition?
Find out which supplements help improve memory, brain function and cognition, including fish oil, some B vitamins, cocoa, and curcumin. ConsumerLab's answer explains the evidence for supplements promoted to help with brain function and cognition.
CL Answer
Which supplements should be taken with food?
Find out which supplements should be taken with food, including vitamins A, D, E, and K, fish oil, and CoQ10.

Product Review
Acai Berry Supplements and Beverages Review
Find the Best Acai Berry Supplements and Beverages. See Which Acai Berry Supplements and Beverages Passed Our Tests of Quality.
Product Review
L-Theanine Supplements Review
Touted for Stress Relief, Do L-Theanine Supplements Work?
Clinical Update
2/27/2018
Matcha Green Tea for Memory & Cognition?
Can drinking matcha green tea enhance memory or have other cognitive benefits? Find out what a recent study showed in the Memory and Cognition section of the Green Tea Review.
Clinical Update
4/24/2018
Avoiding Green Tea Toxicity
The European counterpart to the FDA recently issued its conclusions regarding toxicity from green tea catechins. Get the details in the Concerns and Cautions section of the Green Tea Review.
Clinical Update
3/20/2019
Hot Tea & Cancer
Drinking very hot tea increases the risk of developing esophageal cancer. How long should you wait after brewing to drink tea? Find out what recent studies are showing in the What It Does section of the Green Tea Review. Also see other tips for the best way to prepare brewable green tea, and our Top Picks among green tea products.
Clinical Update
10/27/2018
Cleaner Green Tea
A recent study found that it is possible to modestly reduce pesticides in green tea by following a certain procedure before brewing. For details, see the How to Take section of the Green Tea Review. (Also see our Top Picks among green teas, matcha powders, and supplements.)
Clinical Update
10/06/2017
Green Tea & Prostate Cancer
A recent study investigated the effects of green tea extract in men with pre-cancerous changes of the prostate. Find out if it helped in the "What It Does" section of the Green Tea Supplements and Drinks Review >>
Clinical Update
2/07/2017
Colon Polyps and Green Tea
A study of middle-aged men and women found that those given green tea extract after colon polyp removal were much less likely to develop additional polyps than those who did not take the extract. For details about the extract and dose, see the "What It Does" section of the Green Tea Supplements Review (which includes our tests of supplements and teas) >>
Clinical Update
6/02/2013
Don't Drink Too Much Tea!
After publication of our Green Tea Review last month, a CL member emailed us about the potential danger of getting too much fluoride from tea. Although this won't happen from just a few cups of tea daily, there have been recent cases where habitually drinking extremely large amounts of tea has caused "fluorosis" causing bone pain and even tooth loss. See the warning (and our tests of green tea products) in the updated Green Tea Review >>
Clinical Update
9/25/2011
Green Tea Extract and Weight Loss
A new study using a high dose of green tea extract showed it to help sedentary overweight men lose weight, while those taking placebo gained weight. This is not the first study to show this potential benefit of green tea extract. The effect may be due to the compound EGCG. Get the facts, including how much EGCG was in the product, in the Weight Loss Supplements Review. More >>
Clinical Update
1/27/2024
Green Tea Interaction With Medicine
Drinking green tea may significantly affect blood levels of a common beta-blocker, as well as statins, blood-thinners, and thyroid medication. Be aware of these interactions. See the Concerns and Cautions section of our Green Tea Review.
Clinical Update
8/04/2023
Chemical Solvents in Tea?
Are chemical solvents used to decaffeinate green tea? Find out in our Green Tea Review, which includes our Top Picks for green tea. Also learn about solvents in coffee.
Clinical Update
9/12/2024
Matcha for Sleep?
Does taking green tea matcha powder before bedtime improve sleep? Find out what a recent study showed in the Sleep section of our Green Tea Supplements Review, which includes our Top Picks among matcha powders and other green tea products.
Clinical Update
5/19/2025
Lead in Matcha Green Tea
An independent group recently tested 9 matcha green tea powders and reported that levels of lead and other heavy metals far exceeded "Action Levels." Get our take on this and how the results to compare our own in our Green Tea Review, which includes our Top Picks for matcha and other green teas.
News Release
November 02, 2023
Spirulina and “Greens” Supplement Tests Show Some Problems
White Plains, New York, November 2, 2023 — ConsumerLab tests of spirulina, chlorella, and “greens” supplements revealed that tablets of three brands of spirulina failed to disintegrate within the allotted time, and several other products contained amounts of lead unsuitable for regular consumption ...
News Release
March 09, 2023
L-Theanine Supplements Reviewed by ConsumerLab. Top Pick Chosen.
White Plains, New York, March 9, 2023 —L-theanine supplements are often promoted to reduce stress and improve sleep and cognition.
Product Review
Lavender and Tea Tree Essential Oils Review
Oils Tested for Authenticity and Purity

CL Answer
Does Gynostemma pentaphyllum really work as an AMPK activator and help for diabetes, high cholesterol, or weight loss?
Find out if Gynostemma pentaphyllum ( jiaogulan) works as an AMPK activator and helps to lower blood sugar and treat diabetes. ConsumerLab.com's answer explains.

Product Review
Turmeric and Curcumin Supplements and Spices Review
See Our Top Picks Among Turmeric Products

CL Answer
Which foods and supplements contain lead, cadmium, or arsenic?
Discover which foods and dietary supplements contain lead and other metals, the limits, who’s testing, and how to stay safe.

CL Answer
I take levothyroxine (Synthroid), a thyroid hormone to treat hypothyroidism. Are there supplements or foods I should avoid, or be taking, due to this drug?
Is taking certain supplements and hypothyroidism medications bad? Learn which supplements can interact with thyroid hormone medications like levothyroxine (Synthroid), thyroid hormone levels, and laboratory tests.
Product Review
Olive Leaf Extract Supplements Review
Learn What Olive Leaf Does and the Best Brands

Product Review
Ginseng Supplements Review
Find the Best Ginseng Supplement. Key "Ginsenosides" Found to Range 10-Fold Across Products.

News Release
February 24, 2023
Probiotics Rise in Popularity as Vitamin C, Melatonin, and Others Dip in Latest ConsumerLab Survey of Supplement Users
White Plains, New York, February 25, 2023 —A recent survey of 8,600 people who regularly use dietary supplements shows that probiotics (+3.04 percentage points), quercetin (+2.3 pts), and vitamin K (+1.
Product Review
Lion's Mane and Chaga Supplements Review
Read Labels Carefully -- Many Can Mislead

Product Review
Electrolytes & Sports Drinks Review
See Which Sports Drinks, Powders, and Pills Deliver the Right Electrolytes
CL Answer
How does Athletic Greens’ AG1 compare to multivitamins and Greens powders? Is AG1 worth it?
ConsumerLab reviews the ingredients in AG1, its safety and possible side effects, and how AG1 compares to other products.

CL Answer
Are supplements which claim increased absorption or improved bioavailability telling the truth? Is it worth paying more for these? Are there concerns?
Learn more about bioavailability enhancers, including emulsifiers and enzyme inhibitors.

CL Answer
Which vitamins and supplements are good for acne, and are there any that make it worse?
Information about supplements for acne and clearer skin. Plus, find out which vitamins and supplements can make acne worse.

Clinical Update
3/14/2023
Are Plastic Tea Bags Okay?
Are plastic (nylon) tea bags okay to use? There are two potential problems, as we explain in the ConsumerTips section of our Green Tea Review. Also see our Top Picks for green tea.
Clinical Update
9/02/2022
Black Tea for Heart Health?
Drinking green tea has been linked with reduced risk of death due to heart disease, but how about black tea? See what a recent study found in the Cardiovascular Disease section of our Green Tea Supplements and Drinks Review.
Clinical Update
2/21/2024
Forever Chemicals in Tea?
A recently published study found an association between intake of "unsweetened" tea and higher blood levels of PFAS ("forever chemicals"). Should you be concerned? See the details in the Concerns and Cautions section of our Green Tea Review, which includes our recent Top Picks among products.
Also find out if "plant-based" plastic tea bags may be less likely to release microplastics than regular plastic or nylon tea bags.
A new video explaining how to choose the best green tea is also now available.
Clinical Update
2/27/2024
Matcha Green Tea
We recently announced our Top Pick for "ceremonial" matcha in our Green Tea Review. A CL member asked if we’ve tested "culinary matcha." Although we did not this year, we did in 2021 and we have added those results to the current report.
Clinical Update
1/14/2020
Tea & Heart Disease
Another study shows that people who drink tea are less likely to suffer from cardiovascular disease. Find out how much and what type of tea was associated with benefits in the Cardiovascular disease section of the Green Tea Review. Also see our Top Picks for green tea.
Clinical Update
Clinical Update
1/13/2025
Plastic from Tea Bags
A small study suggested that some plastic tea bags release much larger amounts of nanoplastics than others. For details, and how to avoid nanoplastics from tea bags, see our Green Tea Review, which includes our Top Picks for green tea.
Clinical Update
6/16/2025
Bigelow Tea False Claim
A jury recently awarded consumers of certain Bigelow teas $2.3 million after finding a "Manufactured in the USA" claim to be false. Get the details in our Green Tea Review, which includes our Top Picks among popular green tea bags, loose tea, matcha tea, and bottled tea.
Clinical Update
3/31/2025
Plastic in Tetley Tea Bags?
Find out if Tetley tea bags contain plastic in the Concerns with Plastic Tea Bags section of our Green Tea Review, which includes information about plastics in tea bags from Bigelow, Numi, Republic of Tea, Tazo, Trader Joe’s, and Twinings.
Also see our Top Picks for green tea (including tea bags, loose leaf tea, bottled teas, matcha, and supplements).
CL Answer
Is it safe to drink coffee regularly? Does drinking coffee increase or decrease the risk of cancer?
Learn more about coffee, its safety, if it's good for you, and if it impacts your risk of cancer.

CL Answer
Does drinking coffee deplete vitamins or minerals, such as magnesium, in the body? Can adding magnesium or other minerals reduce the acidity or improve the flavor of coffee?
Coffee can reduce absorption or increase loss of certain vitamins and minerals. Find out which are affected, and what steps can be taken to reduce the effect of coffee on these nutrients.

News Release
August 10, 2022
Best Lavender and Tea Tree Essential Oils Identified by ConsumerLab
White Plains, New York, August 10, 2022 — Lavender essential oil as aromatherapy may reduce anxiety and tea tree essential oil is used topically for various skin ailments.
CL Answer
Dietary Supplements and Herbs to Avoid When Breastfeeding
Find out which supplements should be avoided when breastfeeding, including medicinal herbs, soy, and flaxseed.

CL Answer
Can any vitamins or minerals help prevent or reduce canker sores or cold sores?
Find out which, if any, supplements, including iron, L-lysine, monolaurin, olive leaf extract, vitamin B-12, and zinc, may be beneficial for canker sores or cold sores.
Product Review
Amla Supplements Review
Only 3 of 8 Amla Powders and Pills Pass CL's Tests

CL Answer
With herbal supplements, what is the difference between root powder and root extract? Does it matter?
Learn more about the different forms of herbal supplements, including aloe, boswellia, ginkgo, green tea, and milk thistle.
CL Answer
What is Protandim and does it really work?
What is Protandim and does it really work? Can Protandim reduce oxidative stress? ConsumerLab's answers discusses Protandim's effectiveness, reviews and studies.

CL Answer
How much caffeine is really in dark chocolate bars?
Just how much caffeine is really in chocolate bars? Get our test results on the caffeine content in dark chocolate and cocoa powder.

Recalls & Warnings
November 21, 2017
Health Canada Calls for Stronger Warnings of Liver Risk on Green Tea Extract Products
On November 15, 2017, Health Canada (the Canadian equivalent of the FDA) recommended stronger warning statements about the risk of liver toxicity on labels of supplements containing green tea extract sold in Canada.
Product Review
Pomegranate Juice and Supplements Review Article
What Are the Benefits of Pomegranate Juice and Supplements? Find Out What Pomegranate Can and Cannot Do For Your Health.

CL Answer
Can drinking coffee weaken bones or make arthritis worse?
Learn about the effects of coffee intake on bone mineral density, risk of osteoporosis, and risk of fracture. Also, find out if drinking coffee might increase the risk of arthritis or worsen symptoms.

CL Answer
Which supplements help reduce the risk of colorectal cancer?
Find out which supplements may reduce the risk of colorectal cancer, including folate, fiber, selenium, and vitamin D.
Product Review
Joint Health Supplements Review (Glucosamine, Chondroitin, MSM, Boswellia, Collagen and Turmeric)
18% of Joint Health Supplements Failed to Pass Our Tests. See Which Passed or Failed, and Our Top Picks.

Product Review
Rhodiola Rosea Supplements Review
Do Rhodiola Supplements Help With Depression and Anxiety? Find Out and See Which Rhodiola Supplements Provide the Best Quality & Value.

CL Answer
Do hair loss supplements, such as Viviscal, Hair La Vie, and Nutrafol, or topical essential oils work?
Vitamins and supplements that may help with hair loss and thinning, including saw palmetto, beta-sitosterol, protein, iron, and vitamin D.

CL Answer
I read that sterol supplements to lower cholesterol like CholestOff can block the absorption of vitamins. Is that true?
Find out if CholestOff and other sterol supplements can block vitamin absorption in the body and other safety concerns.

Recalls & Warnings
November 04, 2015
Green Tea from Tea Bags Linked to Acute Hepatitis
According to a case report published on September 23, 2015 (Lugg, BMJ Case Rep 2015), a 16-year old girl in England developed acute hepatitis in connection with the consumption of green tea.
CL Answer
Can fluoride in toothpaste, mouth rinses or drinking water cause attention-deficit/hyperactivity disorder (ADHD) or other neurodevelopmental disorders in children?
Find out if community water fluoridation increases the risk of attention-deficit/hyperactivity disorder (ADHD) or causes lower IQ in children, and learn what the American Dental Association recommends when using fluoride-containing toothpaste and mouth rinses in children.

CL Answer
Are there supplements I should take, or take differently, when intermittent fasting?
Supplements that may help when intermittent fasting. Information about whey protein, CoQ10, magnesium and others, plus coffee, fiber and more.

CL Answer
I take warfarin (Coumadin), an anticoagulant drug. Are there supplements I should avoid, or be taking, due to this drug?
Find out which supplements should be avoided when taking blood thinners (anticoagulants) like warfarin (Coumadin), including vitamin K, St. John's wort, and curcumin.

CL Answer
When taking a statin drug like Lipitor or Crestor, are there supplements I should avoid or take?
Learn about the interactions between certain supplements and atorvastatin (Lipitor), rosuvastatin (Crestor), and other cholesterol-lowering statins.
Product Review
Iron Supplements Review (Iron Pills, Liquids and Chews)
See Which Iron Supplements Are CL's Top Picks for Different Needs

Clinical Update
8/25/2023
Supplements for Memory
Find out if ingredients such as apoaequorin (found in Prevagen), ashwagandha, coffee extract, ginger, green tea, phosphatidylserine, or vitamin E, help improve memory and cognition in our article about supplements and foods for memory.
Clinical Update
10/05/2019
L-Theanine for Sleep and Cognition?
Several studies have evaluated whether a single dose of L-theanine (found in green tea) can reduce stress and anxiety. A new study has evaluated the effect of L-theanine when taken daily for several weeks. See the results in the What It Does section of the L-Theanine Supplements Review. Also see our top choices for L-Theanine.
Clinical Update
4/27/2019
No Ginseng In Ginseng Tea?
A popular maker of bottled teas is being sued for allegedly not putting ginseng in its ginseng green tea. For details, see the Quality Concerns section of the Ginseng Supplements Review. Also see which ginseng products were Approved by ConsumerLab (and see our tests of various Green Teas).
Clinical Update
3/18/2017
Tea Drinking Associated with Less Cognitive Decline
A new study found tea drinkers to have significantly less cognitive decline than others. Read the details in the "What It Does" section of the Green Tea Supplements Review (which includes our tests of teas) >>
Clinical Update
11/14/2017
How Much Tea Is Safe?
Green and black teas contain fluoride, which helps build teeth and bones, but excessive intake of teas can cause teeth and bones to become brittle ("fluorosis"). How many cups of tea are safe? Find out now based on recent tests of fluoride in green, black, and herbal teas in the "Concerns and Cautions" section of the Green Tea Review >>
Clinical Update
10/14/2024
Rooibos Tea?
What are the potential health benefits of rooibos tea, how does it compare to black and green tea, and is it safe? Find out in our article about rooibos tea.
Clinical Update
10/21/2024
Benefits of Mullein Tea?
What is mullein tea and does it really reduce coughing, or help ward off the flu or other viruses? Find out in our new article about the potential health benefits and safety of mullein tea.
Also see our Green Tea Review, including our Top Picks.
Clinical Update
3/27/2025
Plastic in Trader Joe's Tea Bags?
We asked Trader Joe's if there is plastic in its green tea bags. See Trader Joe's response as well as those of Bigelow, Numi, Republic of Tea, Tazo, and Twinings.
Clinical Update
4/21/2025
Are Plastics in Tea Bags Safe?
A plant-based plastic known as PLA is used in some tea bags, but a new study in animals suggests ingesting PLA might have adverse effects. Get the details in the Concerns With Plastic Tea Bags section of our Green Tea Review, which identifies which tea bags include plastic, and which tea companies state they do not use plastic, and see our Top Picks among brewable teas.
Clinical Update
2/10/2025
Avoiding Microplastics from Tea Bags
Recent studies have raised concerns about microplastics released from plastic tea bags. To avoid plastic tea bags, see the Results table in our Green Tea Review, which lists the type of tea bag (paper, plastic, or mixed) for each of the bagged teas we’ve tested. Also see our Top Picks among brewable teas and a list of tea companies that state they do not use plastic in their tea bags.
Clinical Update
2/24/2025
Plastic in Bigelow Tea Bags?
Due to concern over microplastics, we asked Bigelow if their tea bags contain plastic. It turns out that some do and some don’t, as we explain in the Concerns with Plastic Tea Bags section of our Green Tea Review, which includes similar information for other tea brands. The Review also includes our Top Picks among green teas and supplements.
Clinical Update
2/27/2025
Lead & Tea
Although some teas are contaminated with lead (as we have shown), a recent study demonstrated that tea can also remove lead from the water in which is it brewed or steeped. Get the details in the Brewing section of our Green Tea Review, which includes our Top Picks among green teas.
Clinical Update
3/10/2025
Plastic in Twinings or Tazo Tea Bags?
We asked Twinings and Tazo if their tea bags contain plastic. See their responses in the Concerns with Plastic Tea Bags section of our Green Tea Review, which includes similar information for other tea brands. The Review also includes our Top Picks among green teas and supplements.
Clinical Update
3/17/2025
Plastic in Numi Tea Bags?
We asked Numi if their tea bags contain plastic. See their response in the Concerns with Plastic Tea Bags section of our Green Tea Review, which includes similar information for other tea brands. The Review also includes our Top Picks among green teas and supplements.
CL Answer
Does Rooibos Tea Have Health Benefits and Is It Safe?
Learn about the health benefits of rooibos tea, including red and green, unfermented rooibos tea, how is differs from black and green tea, safety, and potential drug interactions with rooibos tea.

CL Answer
Which supplements reduce the risk of stroke? Which increase the risk of stroke?
Find out which supplements (may help reduce the risk of stroke/might increase the risk of stroke.)

CL Answer
Sugar Substitutes: Pros, Cons, and Best Choices
Learn about the pros and cons of using sugar substitutes and artificial sweeteners in place of table sugar. Sweeteners discussed include stevia, monk fruit, and other high-intensity sweeteners; xylitol, erythritol, allulose and other low-calorie sweeteners; and agave syrup, black sugar, coconut sugar, honey, molasses and other sugar alternatives.

CL Answer
Does a USDA Organic seal guarantee that a supplement or natural product has been tested for toxic heavy metals such as lead, cadmium or arsenic?
Find out what the USDA Organic seal on dietary supplements means and whether certified organic supplements and natural products are tested for toxic heavy metals such as lead, cadmium, and arsenic.

Recalls & Warnings
January 18, 2019
Another Green Tea Study Retracted
A study suggesting that green tea catechins reduce the invasive potential of human melanoma cells was recently retracted by the journal PLos One on December 31, 2018 due to concerns over the accuracy of the data.
Recalls & Warnings
February 07, 2011
Maker of Hoodia and Green Tea Products to Pay $2.65 Million Settlement for Unfair Business Practices
On February 1, 2011, supplement maker Irwin Naturals agreed to pay civil penalties, consumer restitution, and and investigative costs to settle a suit in California alleging that Irwin Naturals marketed Hoodia products that did not contain the hoodia gordonii herb despite the product labeling.
Recalls & Warnings
June 27, 2007
Warning Labels to be Required for Green Tea and Black Cohosh Products
On June 26, 2007, the U.S. Pharmacopeia (USP) Dietary Supplements Information Expert Committee (DSI-EC) voted to require cautionary statements on the labels for green tea extracts and black cohosh dietary supplements.
Recalls & Warnings
May 18, 2023
EarthLab, Inc. Warned for Promoting Green Tea, Curcumin, Elderberry & More to Treat Stroke, Cancer & Flu
On April 27, 2023, the FDA issued a warning letter to EarthLab, Inc., dba Wise Woman Herbals, following inspection of the company’s website which found statements about the company’s Green Tea Solid Extract, Curcuma Spp.
CL Answer
Risks of Too Many Vitamins & Supplements
Taking too many vitamin supplements -- or taking too much of a particular supplement -- can have short and long-term adverse effects.

Product Review
Fish Oil, Krill Oil, and Algal Oil Omega-3 (DHA & EPA) Supplements Review
See Our Omega-3 Top Picks and Avoid Rancid Fish Oils.

CL Answer
Is Bottled Water Better Than Tap Water?
Some people consider bottled water to be a favorable alternative to tap water, but is it really better? Find out how bottled water and tap water compare in terms of taste, mineral content, acidity, and contaminants such as microplastics and PFAS.

CL Answer
Do any supplements help with fatty liver disease? Are some diets more beneficial than others?
Find out if supplements such as vitamin E, fish oil, milk thistle, curcumin, choline, probiotics, vitamin D, reishi mushroom, or extra virgin olive oil are beneficial for fatty liver disease and learn which diet (Mediterranean, ketogenic, DASH, or fasting diets) seems to have greatest benefit.
CL Answer
Which supplements can help to lower blood pressure?
Supplements to lower blood pressure, including CoQ10, vitamin C, fish oil, and olive oil, are all explored to find out what works.

News Release
February 24, 2022
Consumers Returned to Pre-Pandemic Supplement Usage in 2021, ConsumerLab Survey Reveals
White Plains, New York, February 24, 2022 —A survey of 8,049 people who use dietary supplements shows many supplements that declined in use in 2020 began bouncing back in 2021, such as magnesium (+2.4 percentage points), and CoQ10 (+2.7 pts).
Product Review
Lycopene Supplements Review
Find the Best Lycopene Supplements. CL Tests Reveal That Not All Lycopene Supplements Contain What They Claim.

Recalls & Warnings
April 25, 2024
Yogi Tea Recalled Due to Pesticides
On April 19, 2024, Yogi Tea issued a recall of over 54,800 packs of Yogi Echinacea Immune Support tea after a sample of echinacea tested above acceptable limits for multiple pesticides. No illnesses have been reported to date.
Product Review
L-Lysine Supplements Review
Read L-Lysine Labels Carefully. Some Can Fool You.
Product Review
D-Mannose Supplements
Find out if D-mannose helps prevent urinary tract infections and how products compare.
Product Review
Selenium Supplements Review
Choose the Best Selenium Supplement. Find Out If You Need Selenium and Which Supplement Is Our Top Pick.
Product Review
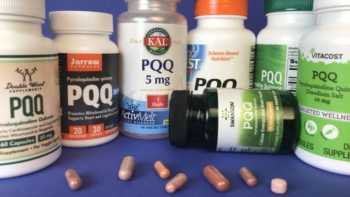
PQQ (Pyrroloquinoline Quinone) Supplements Review
Learn What PQQ Does and Which PQQ Supplements are Best
Product Review
L-Glutamine Supplements Review
Find the Best L-Glutamine Supplements Based on Quality and Value. See our Top Picks.

CL Answer
Is drinking coffee good or bad for heart health?
Learn more about coffee and caffeine, its safety, if it's good for you, how much coffee is too much and its impact on your heart.

Product Review
Green Coffee Bean Extract Supplements Review (for Weight Loss)
Choose the Best Green Coffee Bean Extract. 50% of Green Coffee Bean Extract Supplements Don't Deliver Expected Ingredients.

CL Answer
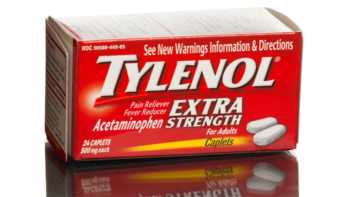
Are there any supplements I should avoid when taking acetaminophen (Tylenol)?
Find out if there are any supplements that should be avoided when taking acetaminophen (Tylenol).
CL Answer
Do any supplements or lifestyle changes reduce the symptoms of tinnitus? Is it true that some supplements can cause tinnitus?
Learn which supplements can ease tinnitus, including melatonin and pine bark extract. Understand which may actually cause tinnitus.

CL Answer
Is Modified Citrus Pectin (MCP) helpful for prostate or breast cancer?
Modified Citrus Pectin (MCP) information including its effects on cancer, such as prostate cancer, and more.

CL Answer
Should you take olive oil as a supplement?
Although extra virgin olive oil has many possible health benefits, such as reduced risk of heart disease and improved blood sugar control, these and other benefits have been demonstrated when olive oil replaces for saturated fats in the diet, not when taken as a supplement, as we explain.

Recalls & Warnings
December 29, 2022
EarthLab, Inc. Warned for Promoting Curcumin, Elderberry to Treat Pain & Flu
On November 10, 2022, the FDA issued a warning letter to EarthLab, Inc., dba Wise Woman Herbals following inspection of the company’s website which found statements about the company’s products to be drug claims because they suggest the products can prevent or treat disease.
Product Review
Glycine Supplements Review
Tests of Glycine Show High-Quality but Major Savings with Right Products.

Product Review
Manuka Honey Review
Find the Best Manuka Honey Based on Quality, Taste, and Value. See our Top Picks.

Product Review
Joint Health Supplements for Pets Review (Glucosamine, Chondroitin, & MSM)
See Which Pet Supplements for Joint Health Passed Our Tests and Which Didn't!

Product Review
Resveratrol Supplements Review (From Red Wine, Knotweed, and Other Sources)
See Which Resveratrol Supplements Were Best In Our Tests and Comparisons. Learn What Resveratrol Can and Can't Do.

Product Review
Water Filter Pitchers Review
See the Best Pitchers For Different Filtering Needs

Product Review
Psyllium Fiber Supplements Review
Find the Best Psyllium Fiber Supplement and Avoid Lead.

CL Answer
Dandelion Tea & Supplements: Health Benefits and Safety
Find out about the potential health benefits of dandelion tea and supplements, as well as safety, side effects, and potential drug interactions.

Recalls & Warnings
November 04, 2016
Liver Injuries Linked With Dietary Supplement Use on the Rise
The number of liver injuries associated with dietary supplement use has increased substantially in recent years, according to a recently published study in the journal Hepatology.
CL Answer
Which supplements reduce hearing loss?
Can supplements such as alpha-lipoic acid, magnesium, N-acetyl-cysteine, folate or vitamins B-12, C and/or E help prevent or improve symptoms among people with hearing loss due to excessive noise or aging? Find out what the clinical evidence shows, and learn whether Advanced Hearing Formula, a supplement marketed for ear health, is likely to help.

CL Answer
Which supplements may benefit people with essential tremor, and which should be avoided?
Learn about supplements used for treating essential tremor, including caprylic acid (also called octanoic acid), thiamin (vitamin B1), cannabidiol (CBD), GABA (gamma-aminobutyric acid), and branched-chain amino acids. Find out which work and which don't. Also find out which supplements and food may make essential tremors worse.

CL Answer
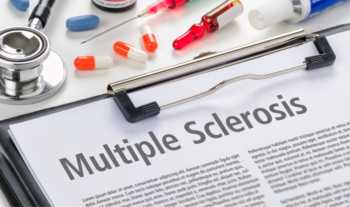
Supplements for multiple sclerosis (MS): Do any supplements reduce symptoms or relapse?
Find out if vitamins or supplements such as ginkgo, fish oil, biotin, vitamins A, C, E, D, and B12, or selenium reduce symptoms or relapse in people with multiple sclerosis (MS).
CL Answer
Can white kidney bean extract lower blood sugar or help me lose weight?
Does white kidney bean extract help promote weight loss? Find out more including evidence from clinical studies and dosage.

Product Review
Nutrition Bars Review (Protein, Energy, and Nut & Fruit Bars)
Our Tests Showed What's Hiding in Protein Bars

News Release
February 09, 2022
Caution With Spirulina Supplements: ConsumerLab Tests Reveal Lead Contamination, Quality Concerns
White Plains, New York, February 9, 2022 — Problems were discovered in three out of four spirulina supplements recently tested by ConsumerLab. One product was found to be contaminated with lead, and two other products, which were tablets, failed to disintegrate within the expected time.
Recalls & Warnings
March 02, 2022
Seller of Holistic Tea Warned by FDA
On February 17, 2022, the FDA issued a warning letter to Vital Health & Wellness following an inspection of the company’s website, which found statements made about the company’s product, Dr. Miller’s Holistic Premium Holy Tea, to be drug claims.
CL Answer
Do any supplements help relieve stress?
Are there actually supplements for stress relief and panic attacks? Learn more about holy basil, fish oil, L-theanine, and ginseng.

CL Answer
Are there vitamins or supplements that can reduce my risk of breast cancer? Do any increase cancer risk?
Find out if certain vitamins, supplements and foods such as fish oil, olive oil, vitamin C and others, can help reduce the risk of breast cancer. ConsumerLab.com's answer explains.

CL Answer
AGZ for Sleep – Is It Worth It?
AGZ claims to "support restful, restorative sleep" without melatonin, but is there evidence to support this use and is it worth its price? Find out.

Product Review
Avocado Oil Review
Find the Best Avocado Oil for Purity, Freshness and Taste. Some Others May Include Rotten Avocado or Other Oils.

Recalls & Warnings
August 07, 2025
Herbalife “Relaxation Tea” Recalled
On July 21, 2025, Herbalife International of America recalled 5,888 units of its Relaxation Tea because an incorrect ingredient was received from the supplier and used in the manufacturing ...
News Release
February 26, 2021
COVID Changed Supplement Popularity in 2020, ConsumerLab Survey Reveals
White Plains, New York, February 26, 2021 — A survey of 9,647 people who use dietary supplements shows that the supplements which experienced the greatest growth in popularity in 2020 were those being promoted to prevent or treat infection with SARS-CoV-2, the coronavirus that causes COVID-19.
Recalls & Warnings
May 02, 2016
The Republic of Tea Recalls Turmeric/Ginger Teas Due to Salmonella Risk
On April 29, 2016 The Republic of Tea issued a recall of several Organic Turmeric Ginger teas because they have the potential to be contaminated with Salmonella.
CL Answer
What are the best supplements for depression and anxiety?
Best natural supplements for depression and anxiety, including the evidence for fish oil, vitamin D, probiotics, and herbal supplements for depression such as curcumin (from turmeric) and saffron.

CL Answer
Do any supplements help for flu?
Which supplements may help prevent or reduce symptoms of colds or the flu, including vitamin D, ginseng, NAC, and elderberry.

CL Answer
What are trace minerals and do I need them?
Trace minerals include boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, zinc, and others. Find out what they do, how much of each trace mineral you should be getting, and who's most likely to be deficient in these minerals.
CL Answer
Which supplements help with arthritis?
Learn more about supplements for joint health, including products such as Wobenzym and Zyflamend and ingredients such as collagen, curcumin, glucosamine and chondroitin, hyaluronic acid, magnesium, omega-3 fatty acids, palmitoylethanolamide (PEA), SAMe, and others.

CL Answer
Mullein Tea: Possible Health Effects and Safety
Mullein tea is often promoted for relieving symptoms of lung conditions, including coughing, hoarseness, sore throat, and shortness of breath. Find out if there is evidence to support these uses, and learn about possible side effects of mullein.

CL Answer
Does tea tree oil work for getting rid of athlete's foot?
Learn more about tea tree oil and its effects on foot fungus (known as athlete’s foot, tinea pedis, or “ringworm” due to the lesion that can occur) and how it can be as effective as some, but not all, over-the-counter antifungal treatments. ConsumerLab explains.

Recalls & Warnings
July 15, 2019
Mislabeled Green Tea Supplement Recalled
On July 11, 2019, DaVinci Laboratories of Vermont issued a recall of 256 bottles of the weight management supplement Dim Plex because they may undeclared fish allergens.
CL Answer
Which supplements and lifestyle changes improve sleep, and which cause insomnia?
Find out which vitamins may help you sleep and learn if any vitamins or supplements can cause insomnia.

CL Answer
I take omeprazole (Prilosec), a proton pump inhibitor, to reduce stomach acid. Are there supplements I should avoid, or be taking, due to this drug?
Supplement interactions with proton pump inhibitor (PPI) drugs like omeprazole (Prilosec) and Nexium.

CL Answer
Is spirulina safe? I heard that it can contain toxins, such as BMAA and microcystin toxin.
Is Spirulina safe? Find out if Spirulina contains toxins like BMAA and microcystin toxin.

CL Answer
Can castor oil eye drops improve or dissolve cataracts? Is castor oil or tea tree oil helpful for other eye conditions such as blepharitis or dry eye?
Can castor oil eye drops help cataracts or for other conditions such as blepharitis? Find out if castor oil eye drops are safe to use, and if they help for other conditions. ConsumerLab.com's answer explains.

CL Answer
A tea called Throat Coat made me lethargic and I was found to have low potassium. Is this a known problem? I was drinking 5 cups a day.
Throat Coat tea information, including licorice root content, potassium loss, and symptoms such as lethargy, increased blood pressure and fluid retention. ConsumerLab.com's answer explains.

CL Answer
Which supplements, foods or diet and lifestyle changes help relieve acid reflux (heartburn), and which worsen it?
Find out which supplements and foods can help relieve acid reflux, and which can make it worse.
CL Answer
5 Tips for Finding a Soap or Body Wash That Won't Irritate Skin
Get 5 tips for finding soap or body wash that won't cause skin irritation. See which soaps we rate best, ingredients to avoid, and phrases on labels that can mislead.

Recalls & Warnings
October 06, 2020
FDA Warns Seller of Red Yeast Rice, Vitamin D, Blood Pressure Supplements, and More
On August 28, 2020, the FDA issued a warning letter to Dr. Sam Robbins, Inc.
News Release
January 31, 2019
Contamination Still an Issue in Some Greens and Whole Food Products, ConsumerLab Tests Reveal
White Plains, New York, January 31, 2019 — Powders and capsules containing ingredients such as wheat grass, spirulina, chlorella, and fruits and vegetables are a popular way to get vitamins, minerals and other plant-based nutrients, but past tests by ConsumerLab have found that these products ...
News Release
May 10, 2018
Big Differences Found Among Green Teas -- ConsumerLab Tests Identify Best Green Tea in Bags, Matcha, Supplements, and Bottles --
White Plains, New York, May 10, 2018 — The benefits of green tea use are associated with the presence of natural polyphenols known as catechins, such as EGCG.
News Release
May 02, 2018
Best Probiotic Supplements and Kombuchas? -- ConsumerLab Tests 41 Popular Products, Reveals Problems and Top Picks
White Plains, New York, May 2, 2018 — With so many different probiotic supplements on the market, each providing different amounts and strains of beneficial bacteria or yeast, choosing the right one can be an overwhelming task.
News Release
February 24, 2018
Magnesium, Curcumin, and Apple Cider Vinegar Rise in Popularity, While Probiotics Dip and Calcium Continues to Decline in Latest ConsumerLab.com Survey of Supplement Users
White Plains, New York, February 24, 2018 — A recent survey of 11,446 people who use dietary supplements shows that magnesium and curcumin and turmeric have leapt into the top 5 most popular dietary supplements, just behind vitamin D, fish oil and CoQ10.
Recalls & Warnings
November 28, 2022
Seller of Elderberry, Tea Warned for Claims of Treating Cold, Flu, Cancer
On October 18, 2022, the FDA issued a warning letter to Rosebud’s Ranch and Garden, LLC after inspection of the company’s website and social media found statements about the company’s Domestic Divas – Colds and Flu (Tea), Domestic Divas - No Pain No Gain Tea, Green ...
Recalls & Warnings
March 01, 2023
Seller of Dr. Miller’s Herbal “Detox” Teas Warned by FDA for Drug Claims
On February 3, 2023, the FDA issued a warning letter to Jackson Health & Wellness Clinic after inspection of the company’s website found statements about the company’s Dr. Miller’s Holy Tea, Dr. Miller’s Super Holy Tea, Dr. Miller’s Ultimate Tea, Dr.
Recalls & Warnings
September 25, 2023
FDA Warns Consumers Not to Use Tapee Tea
On August 31, 2023, the FDA warned consumers not to purchase or use Tapee Tea products after FDA laboratory analysis found them to contain dexamethasone and piroxicam, drugs that are not listed on the product's label.
CL Answer
What is the best way to take curcumin? Is it better to take it all at once or in divided doses?
Find out the best way to take curcumin and turmeric supplements to increase absorption and bioavailability, plus the best way to cook with turmeric spice. ConsumerLab.com's answer explains.

CL Answer
OmegaXL Review: Does It Really Work for Joint and Muscle Pain?
Read our in-depth review of OmegaXL to see if it really helps with joint and muscle pain.
Recalls & Warnings
March 15, 2016
Tea Recalled Due to Salmonella Risk
On March 11, 2016 Awareness Corp. issued a recall of its 7.4 ounce container of Boost Tea because it may be contaminated with Salmonella.
Recalls & Warnings
September 16, 2015
Herbal Extracts Recalled
On September 15, 2015, Iowa Select Herbs, LLC issued a recall of numerous herbal exacts following a permanent injunction which required the company to stop selling supplements.
Recalls & Warnings
October 03, 2014
Seller of Green Tea, Prostate, Pain Supplements and More Warned for Manufacturing Violations and Drug Claims
On July 25, 2014, the FDA issued a warning letter to AMS Health Sciences, LLC following a facility inspection which found the company's products, including saba ACE, UROPOWER, UROSure, Digest-Eze, Shark Cartilage, Colloidal Silver, and Mobilite to be adulterated because they were prepared, packed, ...
Recalls & Warnings
April 26, 2013
Seller of Immune, Cholesterol, Liver and Blood Sugar Products Warned For Drug Claims
On March 20, 2013, the FDA issued a warning letter to Birkdale Medicinals LLC, following a review of the company's website which found statements made about Birkdale products, including Immune Response 247, Cholestat, Silymarin 81 and LevelStat, to be drug claims.
Recalls & Warnings
February 15, 2013
Energy Drink Recalled Due To Bacterial Contamination
On December 21, 2012, NBTY issued a voluntary recall of the energy supplement MET-Rx Extreme Thermo Rage Watermelon 8 fl. oz. (Manufactured by MET-Rx Nutrition, Inc.) due to bacterial contamination.
Recalls & Warnings
January 10, 2013
FDA Warns Aloe and Herbal Supplement Companies For Manufacturing Violations
AloeScience Labs, Inc. -- On November 14, 2012, the FDA issued a warning letter to AloeScience Labs, Inc.
Recalls & Warnings
December 07, 2012
Antioxidant Protandim Supplement Recalled Due To Risk of Metal Fragments
On December 5, 2012, LifeVantage Corporation issued a voluntary recall of dietary supplement Protandim, the Nrf2 Synergizer, due to the potential for contamination with small metal fragments. The recall includes Protandim products sold in the U.S. and Japan between July and November 2012.
Recalls & Warnings
October 24, 2012
Manufacturer of Green Tea, Vitamin E, Omega-3 and Cranberry Supplements Warned For Manufacturing Violations, Misbranding and Drug Claims
On October 16, 2012, the FDA issued a warning letter to dietary supplement manufacturer Advanced Nutritional Technology Inc.
Recalls & Warnings
August 31, 2012
Energy, Fat Loss Supplement Recalled For Ephedrine Alkaloids
On August 28, 2012, dietary supplement re-sale distributor Brand New Energy (BNE) issued a recall of EphBurn 25 after FDA testing found the product to contain ephedrine alkaloids.
Recalls & Warnings
July 13, 2012
Alistrol Health Inc. Warned Over Health Claims on Products
On June 26, 2012, the FDA issued a warning letter to Alistrol Health Inc. for making product statements on the company's websites that constitute drug claims.
CL Answer
What is fermented cod liver oil, such as Blue Ice? Is it better than regular cod liver oil?
Fermented cod liver oil information including if Rosita & Blue Ice (Green Pastures) is as good a source of vitamins and DHA as regular.
CL Answer
How can I avoid aconite – a toxin – in Aconitum supplements?
Aconite is a highly toxic alkaloid found in Aconitum plants ( also called monkshood, wolfsbane, or devil's hood). Aconite poisoning can cause low blood pressure, chest pain, increased or decreased heart rate, and irregular heart rhythm that can lead to cardiac arrest and death. Find out which supplements may contain aconite and how to avoid it.

News Release
February 24, 2017
Vitamin D Supplements Maintain Top Spot in Popularity, as Probiotics Surpass Multivitamins to #4 Behind Fish Oil and CoQ10 in Latest ConsumerLab.com Survey
White Plains, New York, February 24, 2017 — A recent survey of over 9,505 people who use dietary supplements shows the most popular dietary supplement to be vitamin D, followed by fish oil, CoQ10, probiotics, and multivitamins.
News Release
August 08, 2016
ConsumerLab Finds Lead, Cadmium and Arsenic Contamination in Greens and Whole Foods Supplements
White Plains, New York, August 9, 2016 — Powders, pills and drinks made from ingredients like wheat grass, alfalfa, kelp, spirulina, chlorella, leafy vegetables and fruits are a popular choice for people looking to increase their intake of vitamins, minerals and other plant-based nutrients.
News Release
July 26, 2016
36% of Meal Replacement Powders and Shakes Fail ConsumerLab.com's Tests of Quality
White Plains, New York, July 26, 2016 — [Note: This review has been archived and is no longer available online. For ConsumerLab's most recent tests of meal replacement shakes, drinks and mixes, please see our Protein Powders, Shakes, and Meal Replacements Review.
News Release
October 14, 2015
Is Matcha a Better Form of Green Tea? ConsumerLab.com Answers the Question
White Plains, New York, October 14, 2015 — For the past three years, ConsumerLab.com has been testing green tea products, including tea bags, bottled drinks, dietary supplements, and even a K-Cup®.
News Release
March 05, 2015
ConsumerLab.com Shows Chinese Consumers What's Really in Foods and Supplements -- U.S. Testing Company Expands to China, Launches cn.ConsumerLab.com
White Plains, New York, March 5, 2015 — Since 1999, American consumers and doctors have turned to ConsumerLab.com (CL) to find out which nutritional products are highest in quality based independent testing. Today, CL launched a Chinese-language website cn.ConsumerLab.
News Release
July 07, 2014
Which Weight Loss Supplements Are Best? ConsumerLab.com Reviews the Evidence and Tests the Quality of Popular Products
White Plains, New York, July 7, 2014 — Dozens of supplement ingredients have been touted for weight loss, but which have the strongest evidence showing they work and, among those, which products are highest in quality? To answer these questions, ConsumerLab.
News Release
July 08, 2013
Contamination a Common Problem in "Greens" and "Whole Foods" Products According to ConsumerLab.com
White Plains, New York — July 8, 2013 — "Greens" and "whole foods" powders and pills, made from wheat grass, alfalfa, kelp, spirulina, leafy vegetables and other chlorophyll-containing ingredients, as well as fruits, were recently tested by ConsumerLab.
News Release
May 21, 2013
Green Teas Vary in Strength and Amount of Lead Contamination, According to ConsumerLab.com
White Plains, New York, May 21, 2013 — If you drink green tea for your health, be aware that the catechin and caffeine levels can vary by more than 240% across products. Some also contain significant amounts of lead in their tea leaves. This is according to recent tests by ConsumerLab.
News Release
December 21, 2012
ConsumerLab.com Tests Reveal What's Really In Green Tea Supplements and Bottled Drinks
White Plains, New York — December 21, 2012 — How do levels of EGCG and caffeine in green tea supplements and bottled teas compare with levels provided in a typical brewed green tea? It very much depends on the brand, according to new tests by ConsumerLab.com.
News Release
November 06, 2012
50% of Green Coffee Bean Supplements Don't Contain Expected Amounts of Ingredient for Weight Loss, According to ConsumerLab.com
White Plains, New York, November 6, 2012 — Clinical studies suggest green coffee bean extract (GCBE) supplements can aid weight loss, but recent tests by ConsumerLab.com indicate that many popular brands dont contain the amounts of extract listed on their labels.
News Release
February 05, 2012
Fish oil and multivitamins most popular supplements in ConsumerLab.com survey -- Internet most popular place to shop
WHITE PLAINS, NEW YORK — FEBRUARY 5, 2012 — A survey of over 10,000 savvy consumers of supplements shows the most popular supplements to be fish oil, multivitamins, vitamin D, calcium and CoQ10, in that order.
CL Answer
Dihydromyricetin (DHM) – Does It Help and Is it Safe?
Dihydromyricetin (DHM) is promoted for liver health, diabetes, lowering cholesterol, and as a hangover remedy. Find out if it works and whether it is safe.

CL Answer
Is kratom too dangerous to use?
Find out if kratom is safe/legal to use, including potential adverse effects, such as agitation, rapid heartbeat, nausea, drowsiness, seizures, and possible drug interactions.

CL Answer
Can beetroot juice or supplements lower blood pressure or improve exercise performance, and are they safe?
Learn more about beetroot juice and beetroot supplements and if beetroot can really help lower blood pressure.

CL Answer
What are the health benefits of chlorophyll and chlorophyllin, and are they safe?
Chlorophyll supplements are promoted for reducing body odor, preventing cancer, removing toxins from the body, improving digestive conditions and other uses, but do they work and are they safe? Find out and learn if such supplements actually contain chlorophyll.

Recalls & Warnings
December 20, 2021
Pantene, Herbal Essences, & Other Brands of Dry Shampoo and Conditioner Recalled Due to Benzene
On December 17, 2021, Procter & Gamble issued a recall of 26 aerosol spray dry shampoos and conditioners from six brands, including Aussie, Herbal Essences, Hair Food, Old Spice, Pantene, and Waterless Hair Care, because some of the products were found to contain benzene.
CL Answer
Is it true that most coffee is contaminated with mold?
Learn more about mold toxins that may be present in coffee, including mycotoxins, aflatoxin B1, and ochratoxin A (OTA).

CL Answer
Should I be concerned about pyrrolizidine alkaloids in herbal supplements?
Find out which herbal supplement ingredients contain pyrrolizidine alkaloids (PAs), learn why these constituents are a safety concern, and find out how supplements containing these ingredients are regulated in the U.S. and Europe.

Clinical Update
5/01/2025
Tea, Oxalates & Kidney Stones
People prone to kidney stones may be advised to avoid tea due to its oxalate content, but the amount of oxalate in tea varies by the type of tea and how it is prepared. Find out which types of tea contain the most oxalates and ways to reduce the amount of oxalate in tea in our article about kidney stones.
News Release
January 31, 2011
Vitamin D use increased 52% in past two years according to ConsumerLab.com survey of supplement users -- Fish oil continued to surpass multivitamins as most popular supplement and Internet was most common place to buy supplements
WHITE PLAINS, NEW YORK — FEBRUARY 1, 2011 — Among people who use dietary supplements, the most popular products at the end of 2010 were fish oil, multivitamins, vitamin D, calcium and CoQ10 according to a survey by ConsumerLab.com. Use of vitamin D surged in 2010 to 56.
News Release
June 09, 2010
ConsumerLab.com tests "greens" and "whole foods" products; Most offer healthful nutrients, but some found contaminated with heavy metals
WHITE PLAINS, NEW YORK — JUNE 9, 2010 — "Greens" powders, made from wheat grass, alfalfa, kelp, spirulina, leafy vegetables and other chlorophyll-containing ingredients, were recently tested by ConsumerLab.com for potential contamination with heavy metals and pesticides. ConsumerLab.
CL Answer
Which supplements are best for seasonal allergies?
Find out which supplements are best for allergies and can help reduce seasonal allergy symptoms, including bromelain, curcumin, vitamin D, and CLA.

Clinical Update
8/30/2022
Tea Tree Oil Toxicity
Is tea tree oil safe to use around the eye? Be careful -- it can potentially cause eye injury.
There is also concern that tea tree oil can cause growth of breast tissue in young boys. Learn more about this in the Cautions and Concerns section of our Lavender and Tea Tree Essential Oils Review.
Clinical Update
4/14/2023
Skin Reactions to Tea Tree Oil
Topical tea tree oil can cause skin reactions, but there may be ways to minimize the risk, according to a recent review. Get the details in the Cautions and Concerns section of our Lavender and Tea Tree Essential Oils Review.
Also see our Top Pick for tea tree oil.
Clinical Update
1/27/2025
Tea Tree Oil for Cold Sores?
Tea tree oil has been traditionally used for healing cold sores, but does it work? Find out what research suggests in the Tea tree oil section of our Essential Oils Review, which includes our Top Pick for tea tree oil as well as for lavender oil.
Also see: Can any vitamins or minerals help prevent or reduce canker sores or cold sores?
Recalls & Warnings
June 19, 2018
FDA Warns Seller of Weight Control Patches
On June 6, 2018, the FDA issued a warning letter to Health Management Group, Inc.
Recalls & Warnings
January 24, 2013
Food for Health Warned for Manufacturing Violations and Drug Claims on Supplements
On October 5, 2012, the FDA issued a warning letter to Food for Health International, LLC following a facility inspection which found the company's products, including Activz brand Vitamin D, Potassium Iodine, Organic Vitamin C, Whole 9 (a fruit and vegetable meal replacement shake) Control and VMA ...
Recalls & Warnings
July 05, 2013
Seller of Vision Supplement Warned For Drug Claims
On June 25, 2013, the FDA issued a warning letter to Nutrient Synergy, Inc., following a review of the company's website which found statements made about the dietary supplement Nepretin to be drug claims.
Recalls & Warnings
November 30, 2013
"Diuretic" Weight Loss Supplement Recalled
On November 29, 2013, IQ Formulations issued a recall of all lots of diuretic weight loss supplement HYDRAVAX because one lot of the supplement was found to contain an undeclared prescription diuretic drug.
Recalls & Warnings
June 12, 2014
Marketers of Memory Supplement Settle FTC Charges of Deceptive Claims
Martek Biosciences Corporation and i-Health Inc., marketers of BrainStrong Adult, have agreed to settle Federal Trade Commission (FTC) charges that claims the supplement could improve adult memory and prevent cognitive decline were deceptive, and not supported by sufficient clinical evidence.
Recalls & Warnings
January 21, 2014
"Stem Cell" Supplement Recalled Due to Allergen Risk
On January 19, 2014, Stemvida recalled StemAlive 90 Capsules because they were found to contain undeclared milk (labeled as bovine colostrum).
Recalls & Warnings
January 16, 2014
Seller of Tea Supplements and Drinks Warned For Manufacturing Violations and Drug Claims
On December 31, 2013, the FDA issued a warning to Prestige Chinese Teas Company, Inc.
Recalls & Warnings
May 06, 2017
Herbal Teas Recalled Due To Botulism Risk
On May 1, 2017, U.S. Deer Antler Ex. & Imp. of Los Angeles, California, issued a recall of various herbal teas because they have the potential to be contaminated with Clostridium botulinum. This bacterium can cause botulism, a potentially fatal form of food poisoning.
Clinical Update
12/16/2017
Tea & Iron Absorption
A surprising number of foods and beverages, including tea, can interfere with the absorption of iron. When you drink your tea can make a big difference, according to a new study. Get the details in the Iron Supplements Review >>
Clinical Update
5/31/2019
Licorice Tea Caution
Be careful with licorice tea because it can cause serious side effects, as recently happened to a man who drank one to two cups daily. Get more details in our answer about licorice tea.
Clinical Update
11/18/2022
Is Chamomile Tea Safe?
A CL member asked if chamomile tea is safe to drink. It’s generally safe in moderation, but there are concerns with drinking large amounts and with regular consumption during pregnancy. For details, see the Chamomile tea section in our CL Answer about supplements for improving sleep.
Clinical Update
6/19/2025
Licorice Tea Side Effect
Licorice tea, like Throat Coat, can cause low blood levels of potassium – as was recently reported in a woman in New York. Learn more about this, in our article about licorice tea and other licorice products.
Clinical Update
11/07/2023
Are "Greens" Autoimmune Triggers?
Spirulina, chlorella, and alfalfa -- popular ingredients in greens supplements – have been reported to cause, or worsen autoimmune diseases such as lupus, and dermatomyositis. Get the details in the Concerns and Cautions section of our Fruits, Veggies, and Greens Review. Also see our Top Picks among fruits, veggies, and greens supplements.
Recalls & Warnings
March 30, 2021
Real Water Alkaline Water Recalled for Possible Link to Liver Illness
On March 24, 2021, Real Water, Inc. recalled all sizes of Real Water bottled alkaline water because it may be linked to multiple cases of non-viral hepatitis that occurred in Las Vegas, NV in November of 2020.
Clinical Update
10/21/2014
Green Coffee Bean Study Retracted
Evidence for this popular supplement's effect on weight diminished recently when the authors of a clinical study admitted that the data was unreliable. The study had been funded by the maker a popular type of green coffee bean extract. For details, as well as our tests of these products, see the update in the Green Coffee Bean Supplements Review >>
Clinical Update
7/09/2014
Chlorella Lowers Cholesterol and Triglycerides
A daily dose of chlorella (an ingredient included in some "greens" and whole foods supplements) significantly reduced total cholesterol, "bad" VLDL cholesterol, and triglyceride levels in men and women with mildly elevated cholesterol. Get the details, including the dosage used, plus our tests and comparisons of supplements containing chlorella and other "greens" in the updated Greens and Whole Foods Powders and Pills >>
Clinical Update
4/16/2019
Problems With Green Coffee Bean
A recently published analysis found that many green coffee bean extract supplements provided much lower amounts of key compounds than listed – a finding consistent with ConsumerLab's own Review. For details, see the ConsumerTips section of the Green Coffee Bean Extract Supplements Review.
Clinical Update
4/21/2025
Be Careful With AG1
We recently learned that AG1, a popular “greens” supplement, can potentially contain significant amounts of lead and other heavy metals. For details, see the Update in our Greens and Whole Foods Review, which includes how much we found in AG1, and our Top Picks among greens supplements.
Clinical Update
12/13/2024
Liver Concern With Athletic Greens AG1
There have been many reports linking the supplement AG1 by Athletic Greens with liver injury – although these do not prove cause-and-effect. Get the details in the Concerns and Cautions section of our Fruits, Veggies, and Other Greens Supplements Review, which includes our et the details in the Top Picks among products.
Also see: Is there a risk of liver toxicity with certain supplements?
Clinical Update
4/10/2025
Peppermint Tea for Cognition?
Did drinking peppermint tea improve attention or memory in a recent study? Find out in our article about supplements, foods or lifestyle modifications for cognition.
Clinical Update
8/12/2024
Turmeric Tea?
Is turmeric tea a good way to get curcumin (the key compound in turmeric)? Find out in the ConsumerTips section of our Turmeric and Curcumin Supplements Review, which includes our Top Picks among turmeric and curcumin supplements & spices.
Clinical Update
3/28/2023
Pain Reduction with Lavender Oil?
Does spraying the skin with lavender oil reduce the pain of an injection? Find out what a recent study showed in the What It Does section of our Lavender and Tea Tree Essential Oils Review.
Also see our Top Picks among lavender and tea tree oils.
Clinical Update
3/22/2024
Tea Tree Oil Toothpaste?
Dental products containing tea tree oil (such as toothpastes and mouthwashes) can be dangerous. Learn more in our article about dental products, which includes our Top Picks for toothpaste.
Clinical Update
6/20/2023
Teeth Whiteners, Coffee & Tea
Should you avoid coffee and tea after using whitening toothpastes or whitening strips? Find out in the whiteners section of our article about toothpastes and other dental products.
Clinical Update
1/05/2024
Probiotics in Hot Tea?
Some teas now include probiotics, but are probiotics likely to remain viable if steeped in hot water? Find out in the What to Consider When Using section of our Probiotic Supplements Review. Also see our Top Picks among probiotic supplements.
Clinical Update
11/14/2023
Valerian Tea for Insomnia?
Valerian tea is commonly promoted for improving sleep, but does it work? Find out in our Valerian Supplements Review, which includes our Top Picks for valerian.
Also see: Which supplements and lifestyle changes improve sleep, and which cause insomnia?
Clinical Update
8/26/2022
Licorice Concern
Some licorice candies, ice creams and teas may be unsafe due to excessive amounts of the compound glycyrrhizin in these products, according to a recent study. Get the details in our updated article about Throat Coat tea and other licorice-containing products.
Clinical Update
7/12/2022
Toxin in Borage Leaves & Tea
A member asked if borage flower is toxic due to its pyrrolizidine alkaloids. Learn about the safety of borage flower as well as borage leaf (used in teas) and borage oil in our updated CL Answer about pyrrolizidine alkaloids.
Clinical Update
8/06/2021
Caution With Earl Grey Tea and Bergamot
Consuming very large amounts of bergamot (which is found in supplements and teas, such as Earl Grey) may cause adverse effects. Get the details in the Bergamot section of our article about Supplements for Lowering Cholesterol.
News Release
March 02, 2010
ConsumerLab.com finds carcinogenic form of chromium in supplements, including those for weight loss -- Reviews published of supplements containing chromium, green tea, 7-keto DHEA and stimulant formulas
White Plains, New York — March 2, 2010 — A carcinogenic form of chromium, hexavalent chromium, is present is some dietary supplements, according to new tests by ConsumerLab.com.
Clinical Update
9/19/2024
Lowering Cholesterol With Alfalfa?
Did taking alfalfa powder reduce elevated cholesterol levels? Find out what a recent study showed in the What They Do section of our Greens Supplements Review, which includes our Top Picks among "greens" supplements.
Clinical Update
5/12/2025
AG1 Formula Change
Find out what’s recently changed in the popular greens supplement AG1, including a significant increase in a particular vitamin that could potentially cause a side effect. Get the details in our Fruits, Veggies, and Greens Supplements Review, which includes our Top Picks among products.
Clinical Update
3/31/2022
Spinach for Muscle Strength?
Can supplementing with spinach extract improve muscle strength in older adults? Find out what a recent study showed in the What They Do section of our Greens and Whole Food Supplements Review. Also see our Top Picks among greens and whole foods products.
Clinical Update
8/04/2023
Fruits & Veggies in a Capsule?
We said it was too good to be true, and now the distributor of a fruits & veggies supplement will pay over $1 million to settle a false advertising lawsuit. Get the details and our reviews of products sold by Balance of Nature, Juice Plus, Dr. Berg’s, and Nutrilite.
Also see our Top Picks among other greens supplements, including mixed greens powders, chlorella, and spirulina products.
Clinical Update
11/21/2023
Balance of Nature Shut Down
A court ordered the manufacturer and owners of Balance of Nature supplements to stop producing and selling its products. Find out why in our Fruits, Veggies, & Greens Supplements Review, which includes our tests of Balance of Nature, and our Top Picks among fruits, veggies, and greens supplements.
Clinical Update
10/17/2023
Spinach and Weight
Can supplementing with a compound derived from spinach help decrease body weight or improve weight maintenance? Find out in the What They Do section of our Greens Supplements Review, which includes our Top Picks among fruits, veggies, and greens supplements.
Clinical Update
3/06/2015
Green Lipped Mussel Oil Reduces Pain and Inflammation After Exercise
Green-lipped mussel oil (a source of omega-3 fatty acids EPA and DHA) significantly reduced pain and inflammation days after exercise in young healthy men, compared to a placebo, according to a new study. For details, and ConsumerLab.com tests of the product used in the study, see the Fish Oil and Omega-3 Fatty Acid Supplements Review >>
Clinical Update
2/21/2016
Gastrointestinal Side-Effects of Chlorella
In response to a CL Answer about Spirulina, CL members have reported vomiting after using another algae-based supplement, Chlorella — a common ingredient in "greens" powders and drinks. We then reviewed clinical studies of Chlorella and found high rates of other gastrointestinal side-effects. For details see the "Concerns and Cautions" section of the Greens and Whole Foods Powders and Pills Review >>
Clinical Update
8/09/2016
Spirulina Lowers Cholesterol
Spirulina, a common ingredient in greens and whole foods supplements, was found to lower total and "bad" LDL cholesterol, but only in certain people. Get the details, plus our tests of products containing spirulina, in the Greens and Whole Foods Powders, Drinks and Pills Review >>
Recalls & Warnings
August 20, 2013
Weight Loss Supplement Formulas For Men and Women Recalled Due To Undeclared Drugs
On August 16, 2013, Herbal Give Care LLC issued a voluntary recall of all lots of weight loss dietary supplements Esbelder man, Esbelder fem and Esbelder siloutte because they were found to contain the undeclared drugs sibutramine, N-desmethylsibutramine, and N-di-desmethylsibutramine.
Recalls & Warnings
August 01, 2013
Fat Loss and Muscle Enhancement Supplements Found To Contain DMAA
On June 17, 2013, the FDA issued a warning letter to Formulife, Inc., dba Purus Labs, Inc., following a facility inspection which found the dietary supplements Fat Smack XR Thermolipolytic, Muscle Marinade Fresh Fruit and Muscle Marinade Cherry Limeade to contain DMAA.
Recalls & Warnings
November 06, 2014
Seller of Prostate, Heart Supplements and More Warned for Drug Claims
On October 9, 2014, the FDA issued a warning letter to Health Research Laboratories, LLC/New World Health, following a review of the company's websites, which found statements made about AtheChel Advanced, Betarol, BioTherapex, Omega-3 Cardio Plus, RejuvaLifeRx, and Ultimate Health Formula to be ...
Recalls & Warnings
February 21, 2013
FDA and FTC Warn: Supplements Cannot Prevent, Treat Or Cure Cold And Flu
On February 11, 2013, the FDA and the Federal Trade Commission (FTC) jointly issued warning letters to three dietary supplement companies regarding their potential illegal marketing of products to prevent, treat or cure flu virus.
Recalls & Warnings
January 11, 2013
Consumers May Be Eligible For Cold, Allergy and Weight Loss Supplement Refunds
On January 7, 2013, the Federal Trade Commission (FTC) announced that consumers who purchased the Iovate Health dietary supplements Accelis, nanoSLIM, Cold MD, Germ MD, or Allergy MD between January 2006 and July 2010 may qualify for a refund.
Recalls & Warnings
January 04, 2007
Sellers of Popular Weight Loss Supplements Pay $25 Million Over FTC Allegations of Deceptive Advertising
On January 4, 2007, the Federal Trade Commission (FTC) announced that it had filed complaints in four separate cases alleging that weight-loss and weight-control claims were not supported by competent and reliable scientific evidence.
Recalls & Warnings
January 02, 2012
FDA Warns XYMOGEN of Manufacturing and Labeling Violations Regarding Multiple Products
On December 13, 2011, the U.S. FDA sent a Warning Letter to Atlantic Pro Nutrients, Inc. (dba XYMOGEN) regarding labeling and/or manufacturing violations relating to its products, which include Borage CP-240, CoQmax CF, Immune Rx, Iron Glycinate, and Green Tea 600.
Recalls & Warnings
April 14, 2020
Teami Tea Settles Charges of Unproven Claims and Deceptive Celebrity Endorsements
On March 6, 2020, the FTC announced that Teami, LLC, a marketer of tea and skin care products, agreed to settle charges that it was selling products with unproven claims and misleading celebrity endorsements.
Recalls & Warnings
January 24, 2020
Fat-Burning, Energy Supplement Linked to Heart Trouble
A 33-year-old woman in Australia developed cardiac ischemia (a lack of blood flow to the heart) after taking the "fat burning" supplement Alpha Lean-7, according to a recent report in the Journal of Sports Sciences.
Recalls & Warnings
March 18, 2021
Don't Drink Real Water Alkaline Water, FDA Warns After Reports of Liver Illness
On March 16, 2021, the FDA warned consumers not to drink or use Real Water bottled alkaline water while it investigates reports of hepatitis associated with the product.
Clinical Update
5/29/2021
Don't Do This If You Need Iron
If you're low in iron, be aware that many foods dramatically reduce absorption of iron from your diet. Teas can also have a major impact, as shown in a recent study. Get the details in the What to Avoid section of our Iron Supplements Review. Also see our Top Picks among iron supplements.
Clinical Update
9/26/2020
Licorice Danger
A man died after consuming large servings of licorice for several weeks, according to a recent report. Learn about the risks of licorice and the types of products in which it is found in our article about Licorice Root Tea.
Clinical Update
7/02/2019
Licorice Danger
A woman suffered a stroke after consuming a supplement for indigestion that contained a large dose of licorice. Get more details in our answer about licorice tea.
Clinical Update
5/31/2024
Essential Oils Legal Settlement
Young Living Essential Oils has agreed to pay $5 million dollars to settle a class action lawsuit regarding a "therapeutic" claim in marketing its essential oils, including one that we tested in our Lavender and Tea Tree Essential Oils Review. See settlement details in our Recalls & Warnings section.
Clinical Update
8/12/2024
High Blood Pressure from "Liver Support" Supplement
A woman developed high blood pressure after daily use of a “liver support” supplement. Find out the likely reason why in our article about licorice root tea and supplements.
Clinical Update
8/29/2024
Sucralose & Diabetes
Does using sucralose (Splenda) instead of sugar in coffee and tea improve blood sugar control in people with type 2 diabetes? Find out what a recent study showed in our article about sugar substitutes, which includes our Top Picks among sweeteners.
Clinical Update
5/15/2025
Phthalates in Lavender Oil?
Tests by an independent research group found phthalates (plasticizers that can cause hormone disruption) in several popular lavender essential oil products. Find out how these amounts compare to safety limits in the Quality Concerns section of our Lavender and Tea Tree Essential Oils Review, which includes our Top Picks for lavender oil.
Clinical Update
9/20/2022
Best Time to Take Synthroid?
Some people find it difficult to take levothyroxine (Synthroid) in the morning as typically recommended, since this requires delaying breakfast, coffee and tea. Can taking levothyroxine at night, instead, be just as effective? Find out what research shows in our article about supplement and food interactions with levothyroxine.
Clinical Update
7/26/2022
Chamomile for Sleep?
A CL member asked us if chamomile tea is helpful for insomnia. See what research suggests, and learn about potential interactions with chamomile, in our updated answer to the question: What are the best supplements for insomnia? Are there any supplements that interfere with sleep?
Clinical Update
1/27/2024
Licorice & Blood Pressure
Eating black licorice, or drinking tea that contains it, can increase blood pressure and cause heart issues. Get the latest details in our article about licorice.
Clinical Update
2/21/2024
Lavender Oil for Shingles Nerve Pain?
Does lavender oil aromatherapy reduce nerve pain after shingles? Find out what a recent study showed in the Lavender oil section of our Lavender and Tea Tree Essential Oils Review, which includes our Top Picks for lavender oil.
Clinical Update
11/10/2023
Hibiscus for Blood Pressure?
Can supplementing with hibiscus flower in capsule or tea form help lower blood pressure? Find out in our article about supplements for lowering blood pressure.
Clinical Update
1/23/2024
Chamomile and Bleeding Risk?
Does drinking chamomile tea increase bleeding risk? Find out in the chamomile section of our article about supplements for sleep.
Clinical Update
7/14/2023
Best Sweeteners?
Find out which lower-calorie sweeteners are our Top Picks for use in cold beverages, hot beverages (such as in coffee or tea), and baking, in our article about sweeteners.
Clinical Update
7/25/2023
Lavender Oil for Insomnia?
Did the aroma of lavender oil improve sleep or reduce hot flashes in postmenopausal women with insomnia? Find out in the What It Does section of our Lavender and Tea Tree Essential Oils Review, which includes our Top Picks among essential oils.
Also see: Which supplements and lifestyle changes improve sleep, and which cause insomnia?
Clinical Update
10/10/2023
L-theanine for Cognition?
Does taking L-theanine (a compound that occurs naturally in tea) improve attention or cognition in healthy men and women? Find out in the What It Does section of our L-theanine Supplements Review, which includes our Top Picks for L-theanine.
Also see: Do any supplements, foods or lifestyle modifications help with brain function, like memory and cognition?
Clinical Update
4/21/2023
Corn Silk for Nighttime Urination?
Does corn silk tea help reduce nighttime urination? Find out what research suggests in our article about supplements for nighttime urination.
Clinical Update
4/17/2025
Plasticizer in Dark Chocolate, Tuna, AG1
A CL member informed us that one of our Top Picks for dark chocolate was found to contain toxic phthalate compounds in tests by an independent group. See our take on these findings in our Dark Chocolate and Cocoa Review, which includes our Top Picks for dark chocolate and cocoa powder.
Also see the levels of plasticizers found in one of the canned tunas we've tested and the popular "greens" supplement AG1.
Clinical Update
2/13/2025
Juice Plus for Cognition?
Did taking Juice Plus+ improve cognitive function in a recent study? Find out in the What It Does section of our Fruits, Veggies, and Other Greens Supplements Review, which includes our Top Picks among products.
Clinical Update
1/28/2022
B-12 From Chlorella and Spirulina?
A reader asked if chlorella and spirulina are good sources of B-12. The answer is both "yes" and "no," as explained in our Greens and Whole Foods Powders and Pills Review, which includes our recent tests result and Top Picks for chlorella and spirulina supplements.
Clinical Update
12/08/2023
Balance of Nature Back in Business
Balance of Nature has been allowed to resume operations after being shut down for failing to comply with good manufacturing practices. Get our take on Balance of Nature and other products in our Fruits, Veggies, & Greens Supplements Review.
Clinical Update
1/20/2013
Toxin in Algae Supplements
A recently published study found that supplements containing a certain type of algae were all contaminated with a toxin that is a potential carcinogen and can cause liver damage. Get more details in the recent update to the Greens and Whole Foods Powders Review >>
Clinical Update
5/03/2014
Does Fish Oil Help During Pregnancy?
A recent study shows that, at age 4, children born to women given DHA-rich fish oil during pregnancy do not fare better cognitively than other children. Other positive effects around the time of birth, however, suggest that supplementation may be worthwhile. For details, as well as our tests of fish oil supplements, see the Fish Oil and Omega-3 Fatty Acid Supplements Review (Including Krill, Algae, Calamari, Green-lipped Mussel Oil) >>
Clinical Update
6/07/2014
Fish Oil May Reduce Risk of Blood Clots
A study of more than 20,000 people found that those who ate fish several times per week had a reduced risk of venous blood clots, and that getting additional fish oil from supplements reduced the risk even further. Get the details, including our tests of fish oil and other omega-3 supplements in the Fish Oil and Omega-3 Fatty Acid Supplements Review (Including Krill, Algae, Calamari, Green-lipped Mussel Oil) >>
Clinical Update
11/11/2015
Fish Oil Freshness
50% of omega-3 fatty acid supplements recently purchased by researchers in Canada failed tests for freshness, exceeding limits for oxidation. Although the study raises some important issues, it also had significant shortcomings. Get the details in the Fish Oil and Omega-3 Fatty Acid Supplements Review (Including Krill, Algae, Calamari, Green-lipped Mussel Oil) >>
Clinical Update
11/21/2015
Genetically-engineered Salmon: How it Compares
This week the FDA approved a genetically-engineered, farmed salmon as safe for consumption — but how do amounts of omega-3's and hormones compare with non-genetically engineered salmon? Get the details in the update to the Fish Oil and Omega-3 Fatty Acid Supplements Review (Including Krill, Algae, Calamari, Green-lipped Mussel Oil) >>
Clinical Update
6/24/2015
Problem with Too Much Krill Oil
Researchers recently gave overweight men a very large daily amount of krill oil (with a smaller amount of fish oil) with the hope it would improve their insulin sensitivity. Unfortunately, the opposite happened, suggesting that the oil could increase the risk of diabetes and cardiovascular disease. Details about the study, including dosage, are found in the "Concerns and Cautions" section of the Fish Oil and Omega-3 Fatty Acid Supplements Review (Including Krill, Algae, Calamari, Green-lipped Mussel Oil) >>
Clinical Update
3/28/2017
Toxin in Algae Supplements
Some Spirulina supplements and many A. flos-aquae supplements were found to be contaminated with microcystin toxin — a liver toxin, according to a recently published study. For details, see the "Concerns and Cautions" section of the Greens and Whole Food Supplements Review >>
Clinical Update
7/16/2014
Fish Oil May Prevent Cognitive Decline
A study of older individuals found that cognitive function and brain volume (size) were better preserved among those who reported using fish oil supplements than those who did not. The effect, however, was only seen in certain groups of people. For more about the study, and our tests and comparisons of fish oil supplements, see the updated Fish Oil and Omega-3 Fatty Acid Supplements Review (Including Krill, Algae, Calamari, Green-lipped Mussel Oil) >>
Clinical Update
7/16/2014
DHA for Periodontitis (Inflammation Around Teeth)
A new study found that giving DHA (from algal oil) along with low-dose aspirin led to greater reductions in local inflammation and pocketing around teeth than aspirin plus placebo. For more about the study, including dosage, and our tests and comparisons of algal oil supplements, see the Fish Oil and Omega-3 Fatty Acid Supplements Review (Including Krill, Algae, Calamari, Green-lipped Mussel Oil) >>
Clinical Update
3/15/2017
Anti-Inflammatory Effect of Juice Plus+?
Chronic systemic inflammation is a risk factor for cardiovascular disease, diabetes, and cancer. A recent clinical trial assessed the product Juice Plus+ for its ability to reduce this inflammation in obese men and women. The abstract of the published study indicates that it works, but the results tell a different story. For details, see the "What It Does" section of the Greens and Whole Foods Powders, Pills, and Drinks Review (which includes our tests of products) >>
Clinical Update
8/31/2021
Spirulina for Type 2 Diabetes?
Does taking spirulina help control blood sugar levels in people with type 2 diabetes? See what a study found in the What They Do section of our Greens and Whole Food Powders and Capsules Review.
Recalls & Warnings
November 06, 2014
Seller of Cholesterol, Arthritis Supplements and More Warned for Drug Claims
On October 17, 2014, the FDA issued a warning letter to Vitamins Direct (USA) and Golden Pride, Inc., which distribute Flexezy and Physician's Signature brands of dietary supplements, following a facility inspection which found statements made about certain supplements to be drug claims.
Recalls & Warnings
September 23, 2014
Maker of Joint and Weight Products Warned for Hidden Drugs, Manufacturing Violations
On September 15, 2014, the FDA issued a warning letter to West Coast Laboratories, Inc., because the company's joint health supplements, Super ArthGold and Pro ArthMax, were found to contain hidden drugs.
Recalls & Warnings
December 19, 2013
Seller of Sexual Enhancement Supplement and Tea Drink Warned for Drug Ingredients and Drug Claims
On December 5, 2013, the FDA issued a warning letter to Green Planet, Inc. following a facility inspection which found the company's sexual enhancement supplement Night Bullet to contain the drugs sulfohydroxyhomosildenafil, thioaildenafil and aminotadalafil.
Recalls & Warnings
July 12, 2023
FDA Warns Company for Promoting Kratom Products for Pain, Mood, and More
On July 3, 2023, the FDA issued a warning letter to Sunshine Trading Company, Inc.
Clinical Update
7/10/2014
Don't Be Fooled by "Good Source of Omega-3s" Claim
Over the years, ConsumerLab.com has pointed out that products sometimes claim to provide a percentage of the Daily Value (DV) of omega-3 or -6 fatty acids, even though no such Daily Value has been established. Now, the FDA has prohibited certain related statements on omega-3 supplement and food labels that could mislead consumers into thinking a product provides some percentage of this imaginary amount. Get the details, including what to watch out for on labels, plus our tests of fish oil and other omega-3 fatty acid supplements, in the updated Fish Oil and Omega-3 Fatty Acid Supplements Review (Including Krill, Algae, Calamari, Green-lipped Mussel Oil) >>
Clinical Update
1/17/2016
Omega-3's and Cognition
Studies in older individuals have shown that omega-3 fatty acid intake from fish oil may improve certain areas of mental functioning and reduce age-related cognitive decline. Benefits have also been found in younger individuals.
A study has been underway which evaluates the effects of krill oil in adolescents with lower educational performance. It was recently reported that baseline testing (before starting krill) of the students showed a positive association between levels of EPA + DHA in their blood and performance on some, but not all, measures of cognitive performance. Get the details in the Fish Oil and Omega-3 Fatty Acid Supplements Review (Including Krill, Algae, Calamari, Green-lipped Mussel Oil) >>
Recalls & Warnings
August 28, 2013
Tea and Papaya Supplement Company Warned For Drug Claims
On August 12, 2013, the FDA issued a warning letter to Herbal Papaya, LLC, following a review of the company's website and Facebook page which found statements made about Papaya Seed Extract Capsules, 100% Papaya Leaf (Paw Paw Twig) and Papaya Leaf with Rooibos Tea to be drug claims.
Recalls & Warnings
February 28, 2017
Seller of Tea Supplements Warned for Manufacturing Violations
On February 3, 2017, the FDA issued a warning letter to Kumato Labs, following a facility inspection which found the company's products, including CHO WA Tea, COMOXIN IBS Formula and NEMURI Sleep Formula to be adulterated because they were prepared, packed, or held under ...
Recalls & Warnings
May 24, 2013
Brewable Tea and Tea Supplement Company Warned For Manufacturing Violations and Drug Claims
On April 2, 2013, the FDA issued a warning letter to Natures Health Options, LLC, following an investigation which found the company's Charantea Bitter Melon Ampalaya, which is distributed as a dietary supplement in capsule form, and as a tea, to be adulterated because it was prepared, packed, or ...
Recalls & Warnings
February 28, 2013
Maker of Liquid Omega-3 Supplements Warned For Manufacturing Violations
On February 20, 2013, the FDA issued a warning letter to Capco Custom Packaging Inc.
News Release
January 20, 2010
ConsumerLab.com finds quality of most CLA supplements for slimming to be high; One brand mislabeled -- Report is first published in series on supplements for weight loss and slimming
ConsumerLab.com Finds Quality of Most CLA Supplements for Slimming to be High; One Brand Mislabeled
Recalls & Warnings
July 16, 2024
Infla-650 Herbal Dietary Supplement Recalled
On July 16, 2024, Guru Inc. issued a recall of one lot of Infla-650 Herbal Dietary Capsules because they were found to contain acetaminophen, diclofenac, and phenylbutazone.
News Release
April 28, 2009
40% of Green Tea and Selenium Products Fail ConsumerLab.com Review of Cancer-Prevention Supplements; Lycopene Supplements Pass
WHITE PLAINS, NEW YORK — APRIL 28, 2009 — ConsumerLab.com announced results today of its Product Review of Supplements for Cancer Prevention. Among the products that ConsumerLab.com selected for testing, quality problems were found with two out of five green tea supplements.
News Release
March 31, 2009
ConsumerLab.com Warns of Problems with Multivitamins and Vitamin Water -- Defects Found in Over 30% of Supplements Selected for Testing; Several Popular Children's Multis Exceed Tolerable Upper Intake Levels
WHITE PLAINS, NEW YORK — MARCH 31, 2009 — Over 30% of multivitamins recently selected for testing by ConsumerLab.com were found to contain significantly more or less ingredient than claimed or were contaminated with lead.
News Release
April 26, 2006
ConsumerLab.com tests cancer-prevention supplements; several contaminated with lead or low in ingredient — Report available for green tea, selenium, and lycopene supplements
WESTCHESTER COUNTY, NEW YORK — APRIL 26, 2006 ConsumerLab.com announced results today from its new Product Review of Supplements for Cancer Prevention covering 22 products. Cancer is the second leading cause of death in the U.S.
News Release
March 29, 2006
ConsumerLab.com reports on 29 popular melatonin sleep supplements
WESTCHESTER COUNTY, NEW YORK — MARCH 29, 2006 (UPDATED APRIL 7, 2006) — ConsumerLab.com announced test results today from its new Product Review of Melatonin Supplements covering 29 products.
News Release
March 13, 2006
ConsumerLab.com reports on supplements for bone health containing calcium and vitamin D— Results posted for 32 supplements for adults and children; One found contaminated with lead
WESTCHESTER COUNTY, NEW YORK — MARCH 13, 2006 — ConsumerLab.com announced test results today from its new Product Review of Supplements for Bone Health covering 32 adult and children's products containing calcium and vitamin D. Sales of calcium supplements in the U.S.
News Release
January 25, 2006
Sexual enhancement supplements analyzed by Consumerlab.com
WESTCHESTER COUNTY, NEW YORK — JANUARY 25, 2006 — ConsumerLab.com announced today that it found only six out of eleven supplements used for sexual enhancement to contain key ingredients listed on their labels and meet other quality criteria.
News Release
November 15, 2005
Testing by ConsumerLab.com identifies many problems with popular supplements for weight loss, slimming, and blood sugar control
Westchester, NY — November 15, 2005 — In a series of reports released today, ConsumerLab.com revealed test results for supplements used for weight loss, slimming, and blood sugar control. Among the twenty-three products that ConsumerLab.
Recalls & Warnings
March 13, 2025
FTC Sends Refund Checks to Consumers of Pure Green Coffee Supplement
On March 6, 2025, the Federal Trade Commission (FTC) announced it is mailing 39,977 checks totaling more than $905,000 to consumers who purchased Pure Green Coffee, to settle charges the product was promoted with deceptive health claims and marketing practices.
Recalls & Warnings
July 23, 2025
FDA Warns Seller of Allegro Eye Drops, Tea Tree Oil Eyelid Wipes, and More
On July 9, 2025, the FDA issued a Warning Letter to Scope Health Inc.
Recalls & Warnings
February 13, 2019
Supplements Promoted for Alzheimer's Disease and Dementia Sell "False Hope," Warns FDA
On February 11, 2019, the FDA warned consumers to beware supplements promoted to prevent or treat Alzheimer's disease or dementia.
Recalls & Warnings
December 14, 2023
Pain Relief Tea Recalled, Found to Contain Prescription Drugs
On December 13, 2023, WS Global issued a recall of all lots of the company’s Himalayan Pain Relief Tea after products were found to contain diclofenac and dexamethasone, prescription drugs that are not listed on the product’s label.
Recalls & Warnings
September 13, 2014
Marketer Banned From Selling Weight Loss Products
In order to settle FTC charges of deceptive weight loss claims, John Matthew Dwyer III, the co-founder of HealthyLife Sciences, LLC, has agreed to no longer manufacture or market weight loss supplements.
Recalls & Warnings
April 24, 2018
Traditional Medicinals "Throat Coat" Lemon Echinacea Herbal Tea Recalled in Canada
On April 24, 2018, Health Canada (the Canadian equivalent of the FDA) advised consumers that one lot of Traditional Medicinals Throat Coat Lemon Echinacea herbal tea is being recalled in Canada because it has the potential to contaminated by Salmonella.
Recalls & Warnings
January 10, 2018
Lithium Supplement Contaminated With E. Coli, Health Canada Warns
On December 1, 2018, Health Canada (the Canadian equivalent of the FDA) warned consumers that it found Smart Brain Formulations Serotonin Support, a lithium ororate supplement sold on the website Robert Lamberton Consulting, to be contaminated with E. choli (Escherichia coli).
Recalls & Warnings
February 25, 2013
Weight Loss Supplement Recalled Due To Undeclared Drug
On February 21, 2013, Olaax Corp. issued a voluntary, nationwide recall of the company's weight loss supplement MAXILOSS Weight Advanced softgels because they were found to contain undeclared Sibutramine.
Recalls & Warnings
February 14, 2013
Maker of Antioxidant and Anti-Aging Supplements Warned For Manufacturing Violations
On January 10, 2013, the FDA issued a warning letter to dietary supplement manufacturer Consolidated Marketing Unlimited, Inc.
Recalls & Warnings
February 13, 2013
FDA Warns Maker of Weight Loss and Whey Protein Supplements For Manufacturing Violations and Misbranding
January 25, 2013, the FDA issued a warning letter to dietary supplement manufacturer SciLabs Nutraceuticals, following a facility inspection which found the company's dietary supplements and supplement ingredients, including N-Large 2, Flo-Gard AB, Nitrogen Glutamine capsules, Nutritech nutritional ...
Recalls & Warnings
March 03, 2012
Seller of Cancer Cures Warned by FDA
On February 9, 2012, the U.S. FDA sent a Warning Letter to BioAnue Laboratories, Inc. informing it that its websites at www.tumorx.com, www.cancerx.org, www.hopewelltechnologieslimited.com, and www.vmhe.
Recalls & Warnings
September 10, 2012
Maker of Omega-3, Bone and Brain and Supplements Warned For Drug Claims
On August 27, 2012, the FDA issued a warning letter to PruTect Rx indicating that statements on the company's websites about Omega3PruTect, NeuroPruTect, VitaminD3PruTect and OsteoPruTect constitute drug claims, although the products are not approved drugs.
Recalls & Warnings
January 31, 2002
Recall Update for Certain Metabolife Bars - Excessive Vitamin A
The U.S. Food and Drug Administration (FDA) released the following Class I recall information in its January 30, 2002 Enforcement Report.
Recalls & Warnings
December 13, 2016
Case of Hemorrhagic Stroke Linked to Redline Energy Drink
A case of hemorrhagic stroke has been linked to the consumption of a single energy drink, according to a report published on October 11, 2016, in The American Journal of Emergency Medicine.
Recalls & Warnings
August 06, 2016
Some Supplements May Cause or Exacerbate Heart Failure (Includes vitamin E and many herbs)
Taking certain supplements may be dangerous for people with heart failure, according to a statement from the American Heart Association (AHA) which was published in the journal Circulation (August 2, 2016).
Recalls & Warnings
November 12, 2017
Weight Loss and Cleanse Supplements Contain Adulterated and Improperly Labeled Ingredients, FDA Warns
On November 2, 2017, the FDA issued a warning letter to Create-A-Pack Foods, Inc.
Recalls & Warnings
March 17, 2015
Seller of Tea Warned for Drug Claims
On March 4, 2015, the FDA issued a warning letter to Four Elements Organic Herbals, LLC because statements made about HERBAL TEA Power, Energy & Stamina, ORGANIC HERBAL TEA Tulsi TelepaTea, ORGANIC HERBAL TEA To Your Health, Hawthorn Fresh Herb Extract, Elderberry Fresh Herb ...
Recalls & Warnings
March 06, 2015
Seller of Tea Drinks Warned for Drug Claims
On February 26, 2015, the FDA issued a warning letter to the seller of Anamu CancerHerb Tea Box, Anamu CancerHerb Tea Box with Honey and Organic Bottled Tea Drinks because statements made about these products on the websites, www.cancerherbtea.com and www.anamucancerherbtea.
Recalls & Warnings
April 04, 2013
FDA Warns Consumers Of Weight Loss Supplement Containing Undeclared Drug
On April 4, 2013, the FDA warned consumers not to purchase or use the dietary supplement MAXILOSS Weight Advanced Blue because it was found to contain sibutramine.
Recalls & Warnings
March 21, 2013
Probiotic Recalled Due To Undeclared Soy
On March 20, 2013, New Chapter, Inc. issued a voluntary recall of one lot of its Probiotic Elderberry dietary supplement because it may contain undeclared soy.
Recalls & Warnings
September 27, 2013
Company Warned For Distributing Weight Loss Supplement Containing DMAA, Drug Claims
On September 6, 2013, the FDA issued a warning letter to Pure Energy Products, Inc., following a facility inspection which found that the company distributes a weight loss supplement, called obestrim, which contains dimethylamylamine, or DMAA.
Recalls & Warnings
December 19, 2024
Toxic Herb Found in More Weight Loss Supplements
On December 13, 2024, Motivate Me Ashley, LLC recalled the following VidaSlim brand products: VidaSlim 90-day (Original Root, Root Plus, and Root Capsules), VidaSlim 30-day (Original Root, Root Plus, and Root Capsules), VidaSlim 7-day Sample Size (Original Root, Root Plus, and ...
Recalls & Warnings
December 12, 2022
FDA Warns Saffron USA for Promoting Teas to Treat Insomnia, Osteoporosis & Cancer
On September 23, 2022, the FDA issued a warning letter to Saffron USA LLC following inspection of the company’s website which found statements about its Allergy Blend, Chamomile Tea Petals, Diabetic Support Blend, Orange Blast Tea, and Saffron Loose Tea products to be drug ...
Recalls & Warnings
February 06, 2023
FDA Warns Companies Promoting Products to Treat Monkeypox, COVID-19, & Other Viruses
On January 30, 2023, the FDA issued a warning letter to four companies following review of the company websites, which found statements about company products to be drug claims because they were promoted to mitigate, prevent, treat, diagnose, or cure viruses such as monkeypox.
Recalls & Warnings
August 14, 2020
The Green Herb and New Genesis Health Warned for Manufacturing Violations
On July 31, 2019, the FDA issued a warning letter to Davis Ventures, Inc.
Recalls & Warnings
December 06, 2013
Weight Loss Supplement Claims Challenged
On November 27, 2013, the National Advertising Division (NAD) recommended HealthyLife Sciences, LLC, modify or discontinue the use of certain claims made about the company's weight loss supplement Healthe Trim.
Recalls & Warnings
August 21, 2013
Seller of Vitamin, Tea and MSM Supplements Warned For Drug Claims
August 7, 2013, the FDA issued a warning letter to Vibrant Life Vitamins, following a review of the company's websites which found statements made about Taheebo Life Tea, Life Glow Plus, Super Life Glow, Life Glow Basic, Vibrant Life MSM, Herbal MSM, and Organic Germanium to be drug claims.
Recalls & Warnings
June 21, 2005
FTC Settles Claims with Marketers of FiberThin and Propolene
On June 20, 2005, the Federal Trade Commission (FTC) announced that the marketers of the dietary supplements FiberThin and Propolene have settled FTC charges that their misleading weight-loss claims violated federal laws.
Recalls & Warnings
October 24, 2012
FDA Seizes Many Supplements from New York Company Due To Drug Claims
On October 23, 2012, U.S. Marshalls, acting on behalf of the FDA, seized dietary supplements and unapproved drugs from Confidence, Inc., a supplement manufacturer in Port Washington, N.Y. The products included dietary supplements Dr.
Recalls & Warnings
November 05, 2021
Herb Recalled Due to Risk of Lead, Cadmium Contamination
On November 2, 2021, Murray Int'l Trading issued a recall of Herbal Doctor brand Angelicae Sinensis due to potential risk it may contain elevated levels of lead and cadmium.
Recalls & Warnings
March 04, 2022
FTC, FDA, and DOJ Take Joint Action Against Herbal Tea Companies for COVID Claims
On March 3, 2022, the FDA, FTC, and DOJ sued a New York-based marketer of herbal tea, in attempts to permanently block deceptive ads that claim Earth Tea is clinically proven to treat, cure, and prevent COVID-19.
Recalls & Warnings
October 09, 2023
Organifi Settles Charges of Unsupported Claims
Organifi, LLC has agreed to pay $150,000 in civil penalties and $50,000 in investigative costs as restitution to settle charges that the company made unsupported claims that its Gold, Pure, Green Juice, and Red Juice products could balance hormone and cortisol levels, regulate the ...
Recalls & Warnings
November 09, 2023
FDA Warns Biorica International for Cholesterol, Kidney & Cancer Claims
On August 4, 2023, the FDA issued a Warning Letter to Biorica International Corp. following review of the company’s websites and social media, which found statements about the company’s Plaquex Oral Supplements to be drug claims, which are not permitted for dietary supplements.
Recalls & Warnings
May 01, 2024
FDA Warns Consumers Not to Use Ossos-Sans Supplement for Joints
On April 17, 2024, the FDA warned consumers not to purchase or use Ossos-Sans, a supplement promoted to treat osteoarthritis, osteoporosis, and other conditions, because it contains undeclared drugs, including diclofenac and methocarbamol.
Recalls & Warnings
June 24, 2024
Canned Coffee Recalled Due to Botulism Risk
On June 17, 2024, Snapchill LLC recalled its canned coffee products because they have the potential to grow the toxic bacterium Clostridium botulinum.
Recalls & Warnings
August 17, 2022
Seller of Moringa Tea and Other Herbal Products Warned for Drug Claims, Manufacturing Violations
On July 29, 2022, the FDA issued a warning letter to Deggeh Foods, Inc.
Recalls & Warnings
March 15, 2013
Maker of Liquid Supplements Warned For Manufacturing Violations and Drug Claims
On January 31, 2013, the FDA issued a warning letter to Liquid Health, Inc.
Recalls & Warnings
May 04, 2023
FDA Warns MedoLife for Promoting Homeopathic Products to Treat COVID-19, Cancer
On April 19, 2023, the FDA issued a warning letter to Medolife Rx D/B/A MedoLife Corp., AELIA Inc. and Queanta Inc.
Recalls & Warnings
May 24, 2023
Seller of Echinacea, High Blood Pressure Products & More Warned for Drug Claims
On May 12, 2023, the FDA issued a warning letter to H.E.A.L.
Recalls & Warnings
January 04, 2024
Toxic Herb Found in More Tejocote Root Supplements
On January 3, 2024, the FDA warned consumers not to purchase or use certain tejocote root supplements after FDA laboratory analysis confirmed the products contain yellow oleander (Thevetia peruviana), a toxic herb.
Recalls & Warnings
April 03, 2024
FDA Warns Ambaya Gold for Promoting Products for Depression, Cancer, & Arthritis
On December 5, 2023, the FDA issued a Warning Letter to Ambaya Gold Health Products, LLC following review of the company’s website and social media, which found statements about the company’s Brain Balance, Immune System Boost, Dentist In A Bottle, Essensiac, Fulvic Green, Silver ...
Recalls & Warnings
April 25, 2024
Drug Found in Umary Supplement
On April 18, 2024, CTV News in Canada reported tests suggesting that Umary Hyaluronic Acid Dietary Supplement may be adulterated with the prescription anti-inflammatory drug diclofenac, which may explain side effects that have been reported with its use.
Recalls & Warnings
September 05, 2024
Don’t Buy or Use Umary and Amazy Supplements, Warns FDA
On September 5, 2024, the FDA warned consumers not to purchase or use Umary Hyaluronic Acid or Amazy Hyaluronic Acid supplements because testing by the agency found them to contain diclofenac and omeprazole.
Recalls & Warnings
July 22, 2024
Umary Hyaluronic Acid Supplement Recalled
On July 15, 2024, SoloVital.
Recalls & Warnings
March 05, 2021
Seller of Earth Tea Warned for COVID-19 Claims
On February 18, 2021, the FDA issued a warning letter to B4B Corp. following a review of the company's website by the FDA and Federal Trade Commission (FTC) which found the company promoted Earth Tea Extra Strength with unsupported claims that it can treat coronavirus (COVID-19).
Recalls & Warnings
June 13, 2022
Homeopathic Nasal Spray SnoreStop Recalled Due to Microbial Contamination
On June 9, 2022, Green Pharmaceuticals Inc issued a voluntary nationwide recall of one lot of its SnoreStop NasoSpray, a homeopathic nasal spray promoted to reduce snoring, due to the presence of Providencia rettgeri, a bacterium that can cause severe illness in some people.
Recalls & Warnings
August 07, 2021
Poppy Seed Tea, Unwashed Poppy Seeds Pose Risk of Overdose, Death
Unwashed poppy seeds can be contaminated with significant, even dangerous levels of opioid compounds that can cause serious adverse effects, overdose and death.
Recalls & Warnings
November 23, 2022
CBD Not Permitted in Gummies, Candies, Cookies, Candy, or Pet Treats, Says FDA
On November 16, 2022, the FDA issued warning letters to five companies for selling products such as gummies, tea, cookies, lollipops, fruit snacks, hard candies, and pet treats containing cannabidiol (CBD) and/or delta-8 tetrahydrocannabinol (delta-8 THC) as conventional food products.
Recalls & Warnings
January 23, 2020
Flaxseed Salmonella Risk Prompts Recall
Update: (2/4/20) This recall has been expanded to include additional Nopalina flaxseed products, which are highlighted below.
Recalls & Warnings
May 08, 2021
Seller of Immune Bio Green Cell Warned by FDA
On March 30, 2021, the FDA issued a warning letter to Immune & Genetics Protocols, LLC following a review of the company's websites, which found statements made about the company's product Immune Bio Green Cell to be drug claims.
Recalls & Warnings
September 06, 2022
Sand Ginger Powder Recalled in Canada Due to Aconite
On September 1, 2022, Ka Wing Hong issued a recall of Mr. Right brand Keampferia Galanga Powder (sand ginger powder), a common spice used in Asian cuisine, due to aconitine contamination.
Recalls & Warnings
August 25, 2022
White Mulberry Leaf Caused Death of Congressman's Wife, Coroner Reports
The death of Lori McClintock, 61-year-old wife of California congressman Tom McClintock, was caused by dehydration from gastroenteritis caused by “adverse effects of white mulberry leaf ingestion,” according to a coroner's report recently obtained by Kaiser Health News.
Recalls & Warnings
July 06, 2022
FDA Warns Kratom Companies for Arthritis, Pain Relief Claims & More
On January 30, 2022, the FDA issued warning letters to four sellers of kratom products because they were promoted to treat pain relief, opioid withdrawal, blood sugar control, anxiety, and to treat rheumatoid arthritis.
Recalls & Warnings
April 27, 2023
Seller of Magtein, Veggie Caps, & More Warned for Drug Claims, Manufacturing Violations
On March 8, 2023, the FDA issued a warning letter to Spartan Enterprises Inc., dba Watershed Wellness Center, after a review of the company’s website found statements about its Dr. Bob’s Naturals Spirulina, Dr. Bob’s Naturals Magtein, Dr.
Recalls & Warnings
August 16, 2023
FDA Warns Hekma Center, LLC for Promoting Products to Treat Anemia, Diabetes, Depression, & More
On June 2, 2023, the FDA issued a warning letter to Hekma Center, LLC following review of the company’s website and social media, which found statements about the company’s Natural Supplements for Anemia, Lymf (Galium aparine), Natural Supplements for Cardiomyopathy, Magic1 (Moringa ...
Recalls & Warnings
April 26, 2018
Wegmans Recalls Traditional Medicinals Teas Due to Salmonella Risk
On April 17, 2018, Wegmans Food Markets issued a recall of one lot each of Traditional Medicinals Throat Coat Lemon Echinacea herbal tea and Traditional Medicinals EveryDay Detox Lemon tea because they have the potential to contaminated with Salmonella.
Recalls & Warnings
June 23, 2018
Gaia Kratom Recalled Due to Salmonella Risk
On June 21, 2018, Gaia Ethnobotanical, LLC issued a recall of 27 kratom products because they have the potential to be contaminated with Salmonella.
Recalls & Warnings
October 26, 2019
Green Lumber Enhancement Supplements Recalled
On October 22, 2019, GL Holdings issued a recall of Green Lumber, a supplement promoted for male sexual enhancement, because FDA analysis found it to contain tadalafil.
Recalls & Warnings
March 09, 2020
FDA Warns Sellers of Essential Oils, Colloidal Silver & Teas Promoted to Treat Coronavirus
On March 9, 2020, the FDA and FTC announced they have issued joint warning letters to seven companies for selling products such as essential oils, teas and colloidal silver with unsupported claims that they can treat coronavirus (COVID-19).
Recalls & Warnings
January 25, 2021
Clover Leaf Sardines Recalled Due to Risk of Botulism
On January 22, 2021, Clover Leaf Seafoods Corp.
Recalls & Warnings
April 06, 2022
Mandalorian and Mickey Mouse Hand Sanitizers Recalled Due to Benzene, Methanol
On April 1, 2022, Best Brands Consumer Products, Inc.
Recalls & Warnings
May 08, 2018
Badger Botanicals Kratom Recalled
On May 4, 2018, 2018 Badger Botanicals issued a recall of its Green Suma, Red Suma, Green Hulu 2, and Red Hulu 2 kratom dietary supplements because they have the potential to be contaminated with Salmonella.
Recalls & Warnings
March 13, 2020
FDA Finds Problems at 52% of Supplement Manufacturing Sites in U.S. and 42% Abroad
ConsumerLab.com has obtained results of the FDA's inspections in Fiscal Year 2019 (October 1, 2018 - September 30, 2019) of 598 dietary supplement manufacturing facilities in the U.S.
Recalls & Warnings
March 13, 2020
State of Missouri Sues Jim Bakker Show for Selling False Coronavirus Cure
On March 12, 2020, the Missouri Attourney General announced that it sued the Jim Bakker Show for selling a fake coronavirus (COVID-19) cure called Silver Solution.
Recalls & Warnings
August 16, 2018
Kratom Recalled Due to Salmonella Risk
On August 14, 2018, Zakah Life, LLC issued a recall of Super Green Maeng Da Premium Kratom powder, Powerful Red Vein Bali Premium Kratom powder, Super Green Maeng Da Premium Kratom capsules, and Powerful Red Vein Bali Premium Kratom capsules because laboratory testing revealed the presence of ...
Recalls & Warnings
September 12, 2018
FDA Urges Consumers to Avoid Kratom
On September 11, 2018, FDA Commissioner Scott Gottlieb, M.D., urged consumers not to use products containing the kratom, an herb that is often promoted for pain relief and for relieving symptoms of opioid withdrawal.
Recalls & Warnings
June 25, 2019
Kratom Is Dangerous and Not Proven to Treat Addiction, FDA Warns Marketers
On June 25, 2019, the FDA issued warning letter to two marketers and distributors of kratom products, listed below, for illegally selling unapproved, misbranded kratom-containing drug products with unproven claims about their ability to treat or cure opioid addiction and withdrawal symptoms.
Recalls & Warnings
April 17, 2020
Earth Angel Oils Warned for Coronavirus Claims
On April 14, 2020, the FDA issued a warning letter to Earth Angel Oils for selling products with unsupported claims that they can treat coronavirus (COVID-19).
Recalls & Warnings
December 10, 2019
Poisoning from Essential Oils Increasing
Poisonings from essential oil ingestion increased by 16.3% in Australia over a 4-year period, according to a study recently published in the Medical Journal of Australia. The researchers also noted reports of increased frequency and/or severity of essential oil poisonings in the U.S.
Recalls & Warnings
July 10, 2020
Sundial Herbal Recalls More Than Sixty Products Including Turmeric, Cinnamon & Tea
On July 9, 2020, Sundial Herbal Products issued a recall of more than 60 herbal products because they were misbranded and/ or promoted with drug claims. Their labels contain drug claims stating the products can diagnose, cure, mitigate, treat, or prevent disease.
Recalls & Warnings
April 03, 2018
FDA Issues Mandatory Recall of Salmonella-Contaminated Kratom Products
On April 2, 2018, the FDA issued a mandatory recall of all food products containing powdered kratom manufactured, processed, packed, or held by Triangle Pharmanaturals LLC because several of company's products were found to be contaminated with Salmonella.
Recalls & Warnings
April 02, 2018
NutriZone Kratom Supplements Recalled Due to Salmonella Risk
On March 10, 2018, NutriZone, LLC issued a recall of certain kratom-containing powder products because they have the potential to be contaminated with Salmonella.
Recalls & Warnings
October 18, 2017
FDA Warns Seller of Arthritis, Blood Pressure, Diabetes Supplements For Making Drug Claims
On August 15, 2017, the FDA issued a warning letter to Years to Your Health, Inc., following an inspection of the company's website which found a number of their products to be misbranded, as they contained drug claims.
Recalls & Warnings
July 07, 2020
Seller of Homeopathic Products Warned for Manufacturing Violations
On June 19, 2020, the FDA issued a warning letter to Washington Homeopathic Products, Inc.
Recalls & Warnings
June 05, 2020
LifeHealth Science Warned for Manufacturing Violations
On May 15, 2020, the FDA issued a warning letter to LifeHealth Science, which found the company's ORËÁ product to be adulterated because it was prepared, packed, or held under conditions that violate Current Good Manufacturing Practices for dietary supplements.
Recalls & Warnings
April 03, 2020
FDA Warns Pure Plant Essentials for Essential Oil Coronavirus Claims
On April 1, 2020, the FDA issued a warning letter to Health Mastery Systems DBA Pure Plant Essentials for selling products with unsupported claims that they can treat coronavirus (COVID-19).
Recalls & Warnings
April 03, 2020
FDA Warns Seller of Homeopathic Medicine for Coronavirus Claims
On April 1, 2020, the FDA issued a warning letter to Homeomart Indibuy for selling products with unsupported claims that they can treat coronavirus (COVID-19).
Recalls & Warnings
April 03, 2020
FDA Warns Seller of FullerLifeC60 for Coronavirus Claims
On March 30, 2020, the FDA issued a warning letter to FullerLifeC60, LLC, for selling a product with unsupported claims that it can treat coronavirus (COVID-19).
Recalls & Warnings
September 01, 2020
Custom Nutraceuticals, LLC Warned for Manufacturing Violations
On August 6, 2020, the FDA issued a warning letter to Custom Nutraceuticals, LLC, following a facility inspection which found the company's products, including Thermal Revolution Black, Anabolic Blackout Raspberry Lemonade, Militia Re-COMP, and Rhino Rampage Wildberry to be ...
Recalls & Warnings
May 30, 2015
Seller of Omega-3, Probiotics & SuperFoods Warned for Manufacturing Violations, Drug Claims
On May 4, 2015, the FDA issued a warning letter to Dr. Dennis Black, LLC.
Recalls & Warnings
March 11, 2017
Two Hospitalized After Drinking Herbal Tea With Aconite
On March 11, 2017 the San Francisco Department of Public Health announced that two people became ill and required intensive hospital care after consuming tea made from tea leaves purchased from Sun Wing Wo Trading Company in San Francisco.
Recalls & Warnings
August 04, 2014
"Superfood" Greens Recalled Due to Salmonella Risk
On August 1, 2014, New England Greens dba Vibrant Health issued a recall of 10 lots of Green Vibrance and one lot of Rainbow Vibrance because they have the potential to be contaminated with Salmonella.
Recalls & Warnings
September 17, 2003
FDA Warns of Illness from Star Anise Teas
On September 10, 2003, the Food and Drug Administration (FDA) advised consumers not to consume "teas" brewed from star anise.
Recalls & Warnings
July 01, 2013
Teas Recalled Due To Potential Botulism Contamination
One June 28, 2013, Harmony Chai issued a voluntary recall of its Concentrated Black Spiced Chai and Decaffeinated Rooibos Chai bottled tea drinks, because they have the potential to be contaminated with Clostridium botulinum.
Recalls & Warnings
September 14, 2016
Seller of Aloe, Prostate and Joint Supplements Warned for Manufacturing Violations
On April 8, 2016, the FDA issued a warning letter to Salud Natural Entrepreneurs, Inc.
Recalls & Warnings
October 08, 2019
Seller of Tea, Colloidal Silver, Homeopathic Products Warned for Manufacturing Violations
On September 12, 2019, the FDA issued a warning letter to Herbal Healer Academy, Inc.
Recalls & Warnings
June 13, 2013
Seller of Cardio, Immune, Greens Supplements and More Warned For Drug Claims
On May 29, 2013, the FDA issued a warning letter to dietary supplement company Nature's Answer, following a review a the company's website, www.naturesanswer.
Recalls & Warnings
September 09, 2014
Maker of Popular Green Coffee Bean Extract Settles FTC Charges of Unsupported Weight Loss Claims
Applied Food Sciences, Inc., maker of green coffee bean extract GCA, has agreed to pay $3.5 million to settle FTC charges that weight loss claims made about the extract were not supported by scientific evidence.
Recalls & Warnings
May 20, 2014
FTC Charges Seller of Green Coffee Bean with False Weight Loss Claims, Fake Websites
On May 15, 2014, the Federal Trade Commission (FTC) filed a lawsuit against the sellers of Pure Green Coffee for making unsubstantiated weight loss claims and deceiving consumers with fake "news" websites.
Recalls & Warnings
June 04, 2004
FTC Charges Marketers of Two Supplements with False Claims to Cure Range of Diseases
On June 3, 2004 the Federal Trade Commission (FTC) charged marketers of two dietary supplements with falsely claiming that their products can prevent and cure cancer and other diseases. According to the FTC’s complaint, Boston-area marketers Direct Marketing Concepts, Inc. (DMC), ITV Direct, Inc.
Recalls & Warnings
December 22, 2005
Health Canada Warns Consumers Not to Take Chaparral
On December 21, 2005, Health Canada (Canada's health ministry) warned consumers not to ingest the herb chaparral in the form of loose leaves, teas, capsules or bulk herbal products because of the risk of liver and kidney problems.
Recalls & Warnings
May 12, 2018
Seller of Astaxanthin, Cinnamon, Pine Bark and More Warned for Manufacturing Violations
On March 22, 2018, the FDA issued a warning letter to Get The Tea following a facility inspection which found the company's products, including Get the Tea Astaxanthin Max, Get the Tea Colostrum Complete, and Get the Tea Pine Bark Capsules, to be adulterated because they were ...
Recalls & Warnings
January 27, 2015
Green Coffee Bean Supplement Marketer Settles FTC Charges of Deceptive Claims
On January 26, 2015, the FTC announced that Pure Health LLC, Genesis Today, and owner of both companies, Lindsey Duncan, will pay $9 million to settle charges of deceptive weight loss claims made about the companies' green coffee bean extract products.
Recalls & Warnings
October 06, 2005
Deceptive Marketing of “Supreme Greens" -- Settlement with FTC
On October 6, 2005, the Federal Trade Commission (FTC) announced that three individuals and two companies have settled FTC charges over their roles in the deceptive marketing of Supreme Greens, an herbal supplement.
Recalls & Warnings
November 08, 2012
FDA Warns Consumers of "Slimming Coffee"
On November 8, 2012, the FDA warned consumers not to purchase or use Best Share Green Coffee: Brazilian Slimming Coffee after FDA testing revealed the coffee, which was promoted for weight loss, contains the drug sibutramine.
Recalls & Warnings
April 03, 2012
“Japan Rapid Weight Loss Diet Pills Green” Contains Hidden Drug Ingredient
April 3, 2012: The Food and Drug Administration (FDA) is advising consumers not to purchase or use "Japan Rapid Weight Loss Diet Pills Green," a product for weight loss sold on various websites, such as www.amazon.com, and distributed by Xiushentang.
Recalls & Warnings
April 03, 2014
Maker of Green Coffee Bean Extract and Weight Loss Supplement Warned for Manufacturing Violations
On March 13, 2014, the FDA issued a warning letter to Libi Labs, Inc.
Recalls & Warnings
February 05, 2016
Garcinia, Green Coffee Marketers Pay $43 Million to Settle Charges of False Weight Loss Claims
Sale Slash, LLC, has agreed to pay more than $43 million to settle FTC charges it used unsubstantiated claims and fake celebrity endorsements to promote its Premium Green Coffee, Pure Garcinia Cambogia, Premium White Kidney Bean Extract, Pure Forskolin Extract, and Pure Caralluma Fimbriata Extract ...
Recalls & Warnings
August 31, 2009
Court Orders Marketers of Supreme Greens and Coral Calcium to Pay Nearly $70 Million for Consumer Refunds
On August 27, 2009, a federal district court ordered the marketers of two dietary supplements – "Supreme Greens" and "Coral Calcium" – who claimed the products would cure ailments ranging from cancer and Parkinson’s disease to heart disease and autoimmune diseases to pay nearly $70 million for ...
Recalls & Warnings
December 20, 2017
Seller of Supplements for Pain and Allergies Warned for Manufacturing Violations, Drug Claims
On December 13, 2017, the FDA issued a warning letter to GnuPharma Corporation, following a facility inspection which found a number of the company's products, including Relief capsules, Foundation capsules, Aller-geez capsules, Fit capsules, Aller-Geez Tea, Foundation herbal tea and ...
Recalls & Warnings
August 26, 2014
Seller of Immune Supplement Warned for Drug Claims
On August 12, 2014, the FDA issued a warning letter to Ad-Med Biotechnology, following a review of the company's websites and product labels, which found statements made about Immune Active Adult Formula and Immune Active Children's Formula to be drug claims.
Recalls & Warnings
October 18, 2011
FDA Warns Consumers Against Eighteen Weight Loss Products Containing Drugs
On October 18, 2011, the U.S. FDA advised consumers not to purchase or use eighteen different weight loss products found to contain undeclared drug ingredients. The products are listed below with links to more detailed information and photographs.
Recalls & Warnings
May 09, 2015
Marketers Charged with Using Stolen Email Accounts to Promote Weight Loss Supplements Temporarily Shut Down
On May 4, 2015, the FTC announced it has obtained a court order to temporarily halt the operations of weight loss marketers Sale Slash, charging that the company used affiliate marketers to send illegal spam emails and post banner ads online that led consumers to fake news sites.
Recalls & Warnings
March 26, 2015
Green Algae Weight Loss Supplement Contains Hidden Drug
On March 26, 2015, the FDA warned consumers not to buy or use weight loss supplement Green Algae Combination (Crane Beauty) because it was found to contain lorcaserin.
Recalls & Warnings
March 24, 2017
"Micro Greens" Recalled Due to Salmonella Risk
On March 23, 2017, ChloroFields issued a recall of 20 cases of Asian Mix Microgreens because they have the potential to be contaminated with Salmonella.
Recalls & Warnings
August 06, 2013
Maker of Greens and Whole Foods Supplements Warned For Drug Claims, Manufacturing Violations
On July 19, 2013, the FDA issued a warning to ProNatural Nutrition, LLC, following a facility inspection which found that statements made on product labels, including "Healing to cells and tissues," and "Non-toxic antibiotic" to be drug claims.
Recalls & Warnings
August 01, 2013
Maker of Algae Products Warned For Drug Claims, Manufacturing Violations
On March 8, 2013, the FDA issued a warning letter to Algaen Corporation following a facility inspection and website review which found statements made about the company's AlgaBerry products to be drug claims.
Recalls & Warnings
February 20, 2018
Salmonella Outbreak Linked to Kratom Powders, Pills and Teas
On February 20, 2018, the Center for Disease Control (CDC) warned consumers not to use kratom in any form because it could be contaminated with Salmonella. The CDC is currently investigating an outbreak of illness caused by Salmonella infection associated with the use of kratom.
Recalls & Warnings
April 21, 2018
NxtGen Botanicals Maeng Da Kratom Recalled
On April 18, 2018, NGB Corp. of West Jordan, Utah issued a recall of NxtGen Botanicals Maeng Da Kratom labeled bottles of encapsulated product because they have the potential to be contaminated with Salmonella. To date, one illness has been reported to the FDA.
Recalls & Warnings
April 09, 2018
Kratom Powders and Capsules Recalled Due to Salmonella Risk
On April 5, 2018, Club 13 issued a recall of certain kratom powders and capsules because they have the potential to be contaminated with Salmonella.
Recalls & Warnings
July 10, 2018
Blissful Remedies Kratom Recalled Due to Salmonella Risk
On June 30, 2018, Blissful Remedies issued a recall of three kratom products because they have the potential to be contaminated with Salmonella.
Recalls & Warnings
June 19, 2008
FDA Warns Groups to Stop Selling Fake Cancer 'Cures'
On June 17, 2008, the FDA reported that it had sent Warning Letters to 23 U.S. companies and two foreign individuals marketing a wide range of products fraudulently claiming to prevent and cure cancer.
Recalls & Warnings
August 30, 2012
FDA Warns of Medical Claims Made For Numerous Chinese Herbal Products
On August 15, 2012, the FDA warned Dragon Herbs that promotional statements made on the company's website constitute drug claims for twenty herbal products, including CardioPro 2000, Cordyceps, Duanwood Reishi, Gynostemma, Ginseng and Astragalus Combination, Dang Gui and Gelatin, Sweet Relief, ...
Recalls & Warnings
September 29, 2003
FDA Seizes Bogus Dietary Supplement That Claims to Cure Cancer
On September 18, 2003, at the request of the U.S. Food and Drug Administration (FDA), U.S. Marshals seized the dietary supplements, Forticel and Forticel Mix, from Jean’s Greens in Norway, N.Y.
Recalls & Warnings
April 13, 2016
"Super Herbs" Weight Loss Supplement Recalled
On April 11, 2016 Super Herbs issued a recall of all lots of weight loss supplement SUPER HERBS because it was found to contain sibutramine, desmethylsibutramine, and/or phenolphthalein.
Recalls & Warnings
November 11, 2014
Maker of Children's Vitamins, Weight Loss Supplements and More Warned for Manufacturing Violations
On October 30, 2014, the FDA issued a warning letter to VitalHealth Tech, Inc., following a facility inspection which found the company's products, including Kids Mighty Vites tablets, Omni Jr.
Recalls & Warnings
September 26, 2014
Seller Warned for Promoting Essential Oils to Treat Flu, MRSA, Measles and More
On September 22, 2014, the FDA issued a warning to doTERRA International, LLC.
Recalls & Warnings
August 31, 2012
Recent Cases of Lead Poisoning From Ayurvedic Medications from India
Public health officials have reported six cases of lead poisoning among foreign-born women who took Ayurvedic herbal products imported from India. The cases were reported by the New York City Department of Health and Mental Hygiene.
Recalls & Warnings
August 07, 2015
Male Enhancement and Weight Loss Supplement Containing Drugs Recalled
On August 6, 2015 Blue Square Market Inc. issued a recall of the following supplements which were found to contain undeclared drugs:
Recalls & Warnings
June 07, 2012
Recall of Digestive Health Supplement Due to Salmonella Concern
On June 4, 2012, Botanical Laboratories of Ferndale, WA announced the recall of 38 bottles of 33.8 oz Wellese "Digestive 3 in 1 Health " liquid dietary supplement and 275 bottles of 16 oz.
Recalls & Warnings
March 19, 2007
Canada Warns of Weight Loss Supplement with Rx Drug
On March 14, 2007, Health Canada advised consumers not to use MIAOZI Slimming Capsules because they have been found to contain sibutramine, a prescription medication that should only be taken under medical supervision.
Recalls & Warnings
January 21, 2018
Men's Fiber Supplement Recalled
On January 21, 2018, Break Ventures/California Basics recalled of its fiber supplement for men, Zero for Him (150 count) due to possible Salmonella contamination.
Recalls & Warnings
November 28, 2017
Seven Arrested for Selling Fake Olive Oil in Greece
On November 26, 2017, Greek police arrested and charged seven individuals for fraudulently selling large quantities of sunflower oil as olive oil, as reported in the National Post.
Recalls & Warnings
July 18, 2017
Oat Cereal Recalled Due to Listeria Risk
On July 18, 2017, Garden of Light, Inc., recalled Woodstock Organic Matcha Vanilla Oats because of potential contamination with Listeria monocytogenes.
Recalls & Warnings
September 23, 2014
More Carob Powder Products Recalled
On September 22, 2014, Glaser Organic Farms issued a recall of Raw Carob Powder and Carob Fudgy Brownie because they have the potential to be contaminated with Salmonella.
Recalls & Warnings
October 05, 2011
FDA Warns Nutrition Center Inc (Nutri-West) of Manufacturing Violations
The FDA published a Warning Letter dated September 9, 2011 to Nutrition Center Inc. (dba Nutri-West) informing the firm of manufacturing violations found during an inspection of the firm's facility in Douglas, Wyoming in April, 2011.
Recalls & Warnings
March 10, 2018
Kratom Supplements Recalled Due to Salmonella Risk
On March 10, 2018, PDX Aromatics of Portland, Oregon DBA Kraken Kratom, Phytoextractum, and Soul Speciosa issued a recall of certain kratom-containing powder products because they have the potential to be contaminated with Salmonella.
Recalls & Warnings
January 02, 2004
Health Canada Warns Consumers Not to Use Health Products Containing Comfrey
On December 12, Health Canada advised Canadian consumers not to ingest the herb comfrey or any health products that contain comfrey because they might contain a compound called echimidine, which may cause liver damage.






